Academic Editor: Michael Henein
Significant tricuspid regurgitation (TR) is a common finding, affecting about one in twenty-five subjects among the elderly and presenting more frequently in women than in men. This review summarizes data concerning etiology, epidemiology, pathophysiology and management strategies of TR. The tricuspid valve (TV) has a broad anatomical variability. Classically, TR has been distinguished as primary and secondary or functional TR (FTR), with the latter being the most prevalent. FTR is a multifactorial disorder, resulting from maladaptive right ventricular remodeling secondary to pulmonary hypertension or from atrial fibrillation leading to dilation of the right atrium, tricuspid annulus and base of the right ventricle, with pathological TV coaptation. Thus two main types of FTR can be identified: pulmonary hypertension-FTR and idiopathic-FTR, depending on which factor, ventricular or atrial respectively, is the primum movens of the disease. Appreciable evidence suggests that significant TR leads to worsening prognosis regardless of the underlying etiology and should be addressed as a separate therapeutic target. The treatment of TR in patients undergoing left-sided cardiac surgery is well established. Isolated surgical repair of TR is instead rarely performed because patients are often deemed inoperable due to prohibitive risk. Besides, perioperative mortality remains higher than for any other valve. Several transcatheter TV replacement and repair systems, the latter mostly borrowed from percutaneous treatment systems of mitral regurgitation or from TV repair surgical techniques, have shown efficacy and safety when used on the TV. These could provide an effective treatment option for patients not eligible for surgery.
Tricuspid regurgitation (TR) is a highly prevalent echocardiographic finding in the general population [1]. Historically considered as the “forgotten valve disease” by clinician and interventional cardiologists, TR has become a hot topic in cardiovascular interventions over the recent years. If left untreated until severe, as it often occurs, TR correlates with consistent morbidity and mortality, and a variety of surgical and percutaneous treatments have therefore been proposed [2, 3, 4, 5]. Mortality from isolated surgical repair of TR remains higher than that from surgery of any other valve insufficiency and a large number of patients are often deemed not eligible for surgical repair [6, 7]. Recently, transcatheter tricuspid valve (TV) interventions have emerged as an alternative for the treatment of severe TR. Nevertheless, different from the considerable progress achieved in percutaneous treatment of aortic and mitral valve pathologies, many percutaneous treatment options of TR are still under development [8]. Several anatomical and pathophysiological factors, such as the large size of the tricuspid annulus (TA), the “slow flow” of the right heart cavities, the right ventricle trabeculate structure, and the angle of the TA with respect to the superior and inferior venae cavae (SVC, IVC) are major challenges when considering bioprosthesis percutaneous implantation on the TV. The aim of this review is to provide an overview on anatomical factors involved in the pathogenesis of TR and to discuss prevalent causes, incidence, pathophysiological mechanisms and management strategies of TR, with a focus on currently available transcatheter tricuspid valve repair (TTVr) and replacement (TTVR) systems and on the ongoing trials in this field.
Proper function of the TV depends on the integrity of the different components that make up the “tricuspid valve complex”, formed by: tricuspid leaflets, TA, chordae tendineae, papillary muscles, right atrial and ventricular myocardium [9].
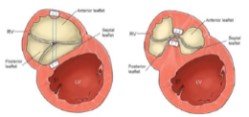
The TV is found in a more apical position than the mitral valve and, anatomically, consists of three leaflets: septal, anterior and posterior (often referred to as the mural or lower leaflet). The anterior leaflet has the largest surface area, while the septal leaflet is the smallest of the three. Notably, the TV leaflet anatomy is highly variable, with fewer than 55% of patients exhibiting the classic 3-leaflet conformation. Recently a new classification of the TV morphology with the use of transesophageal echocardiography (TEE) has been proposed. Based on this classification it’s possible to recognize 4 types of TV morphology: type I, represents the typical 3-leaflet morphology described above. Type II is a 2-leaflet configuration in which the anterior and posterior leaflets are not clearly separated and form a single large leaflet. The type III morphology is characterized by 4 leaflets. This is further subclassified based on the location of the additional leaflet, anterior in type IIIA, posterior in type IIIB, or septal in type IIIC. Lastly, if more than 4 leaflets are present, this morphology is referred to as type IV. The most frequent of these are the type I and IIIB morphologies [10]. The septal leaflet is inserted into a fibrous portion of the TA (near the fibrous trigon). The posterior leaflet is inserted into the lower right ventricle wall, the possible expansion of which is limited by the presence of the diaphragm. These anatomical details have significant pathophysiological implications and explain why TA dilation in functional tricuspid regurgitation (FTR) occurs predominantly along the antero-lateral direction (Fig. 1).
 Fig. 1.
Fig. 1.Typical tricuspid annulus dilation on the antero-lateral side of the tricuspid valve. Dilation typically favors the antero-lateral side as it lacks the supportive anatomy found in the medial and postero-inferior directions. Medially the septal leaflet inserts into a fibrotic section of the annulus while the posterior leaflet inserts into the inferior wall of the right ventricle, which is supported by the underlying diaphragm.
The TA is a complex elliptically-shaped three-dimensional structure in which the postero-septal portion is more apical than the antero-septal one. It differs from the more symmetric “saddle-shaped” mitral annulus. The TA has a dynamic structure, which can vary greatly in size in relation to the different stages of the heart cycle and right ventricle load conditions [11].
The papillary muscles that support the TV are smaller than those that support the mitral, often multiple and variably subdivided. Each papillary muscle connects to the homolateral leaflet via the chordae tendineae (unlike left ventricular [LV] papillary muscles, each of which provides chordae tendineae to each mitral leaflet). This anatomical organization has pathophysiological relevance, as it explains the greater ease with which the TA expands (lacking the resistance to dilation due to the crossing of the chordae tendineae) as a result of right heart cavities enlargement, generating functional valve insufficiency.
The etiology of TR is generally classified according to the presence of an intrinsic TV abnormality (primary TR) and/or the presence of a right ventricular (RV) or right atrial enlargement leading to TA dilation and/or leaflet tethering (secondary or FTR) (Table 1).
| Primary or organic TR | Secondary or functional TR | |||
| Congenital | Left-sided heart disease | |||
| Ebstein’s Anomaly | (MR, mitral stenosis, aortic stenosis) | |||
| Acquired | Pulmonary hypertension | |||
| Direct injury from implantable devices leads | Primary RV dysfunction and dilation | |||
| Rheumatic fever | (RV infarction, arrythmogenic RV cardiomyopathy/dysplasia) | |||
| Carcinoid syndrome | Atriogenic TR | |||
| Right-heart endocarditis | (Atrial fibrillation or isolated right atrium enlargement) | |||
| (iv drug addicts, lead infection) | ||||
| Tricuspid prolapse | ||||
| Marantic endocarditis | ||||
| Connective tissue disorders | ||||
| (Marfan or Ehler-Danlos syndrome) | ||||
| Post-traumatic TR | ||||
| Drug-induced TR | ||||
| Iv, intravenous; MR, mitral regurgitation; RV, right ventricular; TR, tricuspid regurgitation. | ||||
Primary TR is caused by an organic process affecting the valve or the subvalvular apparatus, which can be either congenital or acquired. Congenital TR is generally attributed to Ebstein anomaly, in which the posterior and septal tricuspid leaflets are displaced apically in the right ventricle resulting in its atrialization, and should be suspected in case of TR occurring in young patients [12, 13]. Rheumatic fever remains the most common cause of primary acquired TR worldwide, often presenting with combined stenotic and regurgitant lesions and with a concomitant mitral valve disease [14]. Rheumatic TR can present both with an organic pattern, characterized by the shortening and thickening of tricuspid leaflets leading to imperfect sealing, and a functional pattern, with TA dilation secondary to RV enlargement in patients with left-sided valve disease [15, 16]. A not infrequently cause of primary TR in Western countries is the direct injury from implantable device leads, associated with lead impingement on the leaflets, and/or papillary muscles and chordae tendineae perforation, laceration or transection [17]. In a cohort of 239 implantable cardioverter-defibrillator (ICD) or pacemaker (PM) recipients, almost 40% of subjects developed severe TR within 5 years following implantation [18]. TR is also a common manifestation of carcinoid syndrome, which affects the valves and endocardium of the right heart chambers [19, 20]. In this condition, fibrous plaques are observed on the endocardium surface of tricuspid leaflets, which consequently cannot adequately undergo coaptation [21, 22]. TV infective endocarditis is most frequently seen in intravenous drugs users and in patients with implantable device leads. An echocardiographic finding of severe TR associated with annular abscesses or mobile masses on the leaflets should raise its suspicion [23, 24]. Less common causes of primary TR include: chest wall or deceleration injury trauma [25]; tricuspid prolapse due to myxomatous degeneration, often associated with mitral valve prolapse [26]; connective tissue disorders [27]; systemic lupus erythematosus or rheumatoid arthritis complicated by marantic TV endocarditis [28] and drug-induced TR. Indeed, beyond the higher risk of right-heart endocarditis observed among intravenous drugs users, anorectic drugs (e.g., fenfluramine and phentermine) and the dopamine agonist pergolide have been associated with an increased incidence of TR [29].
FTR develops due to structural alterations in right atrial and/or ventricular myocardial geometry, leading to TA dilation and/or leaflet tethering, both associated with impaired leaflet coaptation. This is by far the most common cause of TR in adults, as shown by echocardiographic studies in which over than 90% patients with severe TR had a functional etiology [30, 31]. Multiple etiologies of FTR have been described. Most commonly, FTR is associated with LV dysfunction and/or left-sided heart disease, leading to left atrial dilation, increased pulmonary wedge pressure and RV afterload [32, 33]. Pulmonary hypertension (PH) arising from other causes than left heart disease, such as primary PH, pulmonary embolism and chronic pulmonary disease, can also cause TR due to increased RV afterload, RV dilation and dysfunction [34]. On the other hand, primary RV diseases, including isolated RV infarction or arrhythmogenic RV dysplasia, occurs less frequently [35, 36]. “Atriogenic” secondary TR is another often underappreciated but frequent cause of FTR. This condition is characterized by isolated atrial enlargement, usually in the presence of chronic atrial fibrillation, with normal right ventricle size and morphology [37].
TR is often diagnosed as an incidental finding on routine echocardiography performed in patients with left-sided heart disease. In particular, up to half of patients with severe mitral regurgitation and one quarter of those with severe aortic stenosis develop a significant (at least moderate) TR [38, 39, 40]. Surgical or endovascular correction of mitral and aortic valve diseases does not seem to limit TR onset or progression. In a recent study including 193 patients with degenerative mitral valve disease undergoing surgery, approximately 20% developed a moderate to severe TR at 2-year follow-up, with mitral valve replacement associated with a two-fold increased risk compared to mitral valve repair [41]. In the setting of aortic valve replacement, more than half of patients with significant TR undergoing transcatheter aortic valve replacement (TAVR) had persistent or worsened TR [42], while surgical aortic valve replacement (SAVR) was associated with greater TR compared to TAVR in the PARTNER-3 (Placement of Aortic Transcatheter Valves-3) trial [43].
Similarly, a 20 to 30% prevalence of significant FTR is observed in patients with both acute and chronic heart failure, which contributes to lower response to optimal medical and device-based therapy [44, 45, 46].
The prevalence of TR in the community is less investigated. The Framingham Heart
Study, a large cardiovascular cohort study, displayed an increased prevalence of
moderate to severe TR among women and the elderly, reaching up to 1.5% and 5.6%
in men and women aged 70 or older, respectively. The prevalence in the entire
cohort was 0.8% [47]. Similarly, in the OxVALVE (Oxford Valvular Heart Disease
Population Study) study, enrolling 2500 subjects aged
The prognostic implications of TR have firstly been assessed in a large
echocardiographic registry of 5223 male patients. After adjustment for several
echocardiographic parameters suggestive of LV and RV dysfunction and PH, severe
TR correlated with an increased mortality at 4-year follow-up (hazard ratio [HR]
1.31, 95% confidence interval [CI] 1.05–1.66, p
Concerning secondary etiologies, the prognostic impact of TR should be stratified according to the presence of treated or untreated mitral and aortic valve diseases, PH, and/or LV dysfunction. The implications of moderate to severe TR associated with mitral regurgitation has been recently evaluated in a substudy of the COAPT (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation) trial, which randomized 614 patients with heart failure with reduced ejection fraction and secondary mitral regurgitation to optimal medical therapy vs. MitraClip. Significant TR was associated with a higher rate of death and/or heart failure hospitalization at 2 years among medically treated patients (HR 1.74, 95% CI 1.24–2.45, p = 0.001) but not in the MitraClip group (HR 1.14, 95% CI 0.71–1.84, p = 0.59) [53]. Severe TR has also been described as an independent predictor of death and worsening heart failure after surgical mitral valve repair in a large US nationwide registry (N = 2952) [54]. In patients with severe aortic stenosis undergoing TAVR in the PARTNER II trial, significant TR was associated with an approximately two-fold increase in one-year mortality, regardless of RV dysfunction severity, and a subsequent meta-analysis confirmed the effect on both in-hospital complications and mid-term survival in this group [55, 56]. On the other hand, in a retrospective study including 226 patients with severe aortic stenosis treated with aortic valve replacement, pre-operative presence of mild or moderate TR correlated with the development of decompensated heart failure and renal dysfunction [57].
Also, in the setting of non-valvular LV systolic dysfunction, there is strong evidence suggesting the prognostic role of TR. A large prospective cohort study (N = 2101) of patients diagnosed with acute heart failure showed that patients with moderate to severe TR (17.2%) had a higher rate of recurrent heart failure hospitalizations [44]. Similarly, among 1421 patients with heart failure with reduced ejection fraction, TR severity was an independent mortality predictor at multivariable analysis (HR 1.55, 95% CI 1.14 to 2.11, p = 0.0325) [58]. In a comprehensive meta-analysis including 32601 patients, mild, moderate and severe TR had a progressively increased risk of all-cause mortality, even after adjustment for pulmonary artery pressures and RV function [59]. Finally, isolated “atriogenic” TR was associated with a lower survival at 10 years, independently from other cardiovascular comorbidities, including the presence of baseline atrial fibrillation versus sinus rhythm [1, 60].
It can therefore be concluded that a significant TR leads to worsening prognosis regardless of the underlying etiology and should be addressed as a separate therapeutic target.
The mechanisms leading to TR are different and heterogeneous for each disease in the case of primary TR. Examples of resulting structural damage are leaflet perforation or restriction, commissural fusion, and chordal tethering or rupture. Primary TR results in pure volume overload of the right heart and therefore is usually associated with annular dilation [61].
FTR is essentially due to right atrium and/or right ventricle dilation with variable degrees of TA dilation and leaflet tethering, according to the underlying cause. FTR is thereby not a valvular disease; it is an abnormality which results from a disease process that primarily affects the atrium or ventricle and subsequently alters the TA size and the mode of TV leaflet coaptation.
Coaptation is the contact surface between leaflets during systole. The greater the coaptation, the less likelihood there is of regurgitation to occur. A physiological coaptation takes place between the body of each leaflet and, therefore, is called body-to-body coaptation. If, for any reason (TA dilation and/or leaflet tethering), the coaptation surface is decreased, then coaptation can take place on the free edge of the leaflets, either symmetrically (edge-to-edge) or asymmetrically (edge-to-body). In these cases, the likelihood of developing TR becomes increasingly high, as according to filling conditions, there is no longer any safety margin to compensate or avoid regurgitation. When significant TA dilation and/or leaflet tethering is present, no leaflet coaptation occurs at any stage of the cardiac cycle, thereby creating a single cavity between the right atrium and right ventricle (Fig. 2). Coaptation is affected by many more variables on the right side, as opposed to the left side. Hence, due to more rapid changes in RV size, shape, and function, the mode of leaflet coaptation is more relevant on the right side. Normal coaptation of the tricuspid leaflets during systole takes place either at the annular level or apically, just below the TA plane, with a good body-to-body coaptation (around 5 to 10 mm of the leaflet bodies in contact with each other). The distance of the point of leaflet coaptation to the plane of TA can be measured and is referred to as the coaptation height. The area contained between the leaflets and the annular plane can be measured and is called the tethering area (Fig. 2).
 Fig. 2.
Fig. 2.Schematic representation of the physiological and pathological modes of leaflet coaptation. (A) Physiological body-to-body coaptation. (B) Edge-to-edge leaflet coaptation (above); edge-to-body leaflet coaptation (below). (C) Lack of leaflet coaptation. CT, chordae tendineae; PM, papillary muscle; RV, right ventricle; TA, tricuspid annulus; TL, tricuspid leaflet.
The mechanisms leading to FTR differ between patients with PH-FTR and the subgroup of patients with FTR in the absence of PH, or other associated cardiac abnormalities that are named as “isolated”, “idiopathic”, or “atrial fibrillation-associated FTR” (Id-FTR). Primary RV dysfunction is quite rare [35, 36], but pathophysiological alterations leading to eventual FTR are similar to those present in case of PH-FTR, since the ventricular factor is the primum movens of the disease.
The most common type of FTR is the one associated with PH. Long-standing PH
causes RV maladaptive remodeling mainly at the expense of the antero-lateral wall
at the mid-ventricular level. This leads to papillary muscles displacement
(bringing the anterior papillary muscle into a more caudal position) and
subsequently TV leaflet tethering. Thus PH-FTR is essentially due to valve
deformation with leaflet tethering and only modest annular enlargement. Valvular
tenting and leaflet tethering are linked to RV elongation and
elliptical/spherical deformation [29, 32, 34]. The RV eccentricity index is
easily measurable as the ratio between the long transverse axis and the short
transverse axis at the mid-ventricular level in short-axis projection. A RV
eccentricity index
 Fig. 3.
Fig. 3.Differences in anatomical and structural findings according to functional tricuspid regurgitation (FTR) pathophysiology: Idiopathic-FTR versus pulmonary hypertension-FTR. On the left side it is depicted Id-FTR pathophysiology. Atrial fibrillation causes the dilation of the right atrium, tricuspid annulus and base of the right ventricle, leading to edge-to-edge leaflet coaptation and then, with progressive annulus dilation, to lack of coaptation. On the right side it is depicted the pathophysiology of pulmonary hypertension-FTR. Right ventricle pressure overload causes the dilation of the right ventricle, with papillary muscles displacement and leaflet tethering. Basal dilation of the right ventricle is initially trivial, then with the progression of leaflet tethering and FTR, there is an overload of the right atrium with subsequent tricuspid annulus dilation and advanced leaflet coaptation defect. ALP, antero-lateral papillary muscle; Id-FTR, Idiopathic-Functional Tricuspid Regurgitation; PH-FTR, pulmonary hypertension-functional tricuspid regurgitation; PMP, postero-medial papillary muscle; RV, right ventricle; SMP, septo-medial papillary muscle; TV, tricuspid valve.
 Fig. 4.
Fig. 4.Transthoracic echocardiographic findings according to functional tricuspid regurgitation (FTR) pathophysiology: Idiopathic (Id)-FTR versus pulmonary hypertension (PH)-FTR. (A) Four-chamber view. (B) Color Doppler in the four-chamber view. (C) Four-chamber right ventricle focused view show a typical Id-FTR in a woman of 74 years, with permanent atrial fibrillation and hypertension. Right ventricle has a conical shape, since only the base is dilated because there is severe right atrium dilation. This results in a leaflet coaptation gap causing severe FTR. There is no leaflet tethering. (D) Four-chamber view. (E) Color Doppler in the four-chamber view. (F) Parasternal window- right ventricular inflow tract view show a typical case of PH-FTR in a man of 87 years, with hypertension, diabetes mellitus and heart failure with reduced ejection fraction secondary to post-infarction ischemic heart disease. Severe right ventricle dilation secondary to pulmonary hypertension leads to papillary muscles displacement and subsequent leaflet tethering. Chronic FTR causes right atrium volume overload and subsequent dilation of the atrium itself and tricuspid annulus, which result in massive FTR.
 Fig. 5.
Fig. 5.Different right heart chambers morphologies. (A) Normal subject, edge-to-edge leaflet coaptation. (B) Primary tricuspid regurgitation due to chordae tendineae rupture, right atrium and ventricle have normal sizes. (C) Idiopathic functional tricuspid regurgitation, with severe tricuspid annulus, right atrium and basal right ventricle dilation. (D) Pulmonary hypertension functional tricuspid regurgitation, the right ventricle is severely dilated, with papillary muscles displacement and leaflet tethering. Tricuspid annulus and right atrium dilation is less evident, at least at the initial stage of the disease.
Id-FTR is related primarily to right atrium, TA and basal right ventricle dilation, with exhaustion of leaflet annular coverage reserve and little or no role for leaflet tethering in the loss of leaflet coaptation. In Id-FTR, basilar dilatation without elongation of the right ventricle results in RV conical deformation (Figs. 3,4,5). Atrial fibrillation, highly prevalent in the elderly, is associated with structural and electrical left and right atrial remodeling and enlargement, being able to explain, at least in part, the anatomical substrates for Id-FTR [60, 63]. In addition, atrial fibrillation is highly prevalent in patients with PH and may also arise as a consequence of PH-FTR, so that the mechanisms aforementioned often overlap in the same patient.
Another important aspect in the pathophysiology of TR is the load dependance. Small changes in RV preload induced by inspiration may cause a 10% increase in RV area and annular size, a 20% change in tethering, but more than doubling of TR effective regurgitant area and regurgitant volume [64]. This is the pathophysiological basis for the extreme changes seen in TR severity with trivial changes in fluid status and preload, and represents a paramount aspect that must be taken into account when evaluating a patient with TR.
Echocardiography, either transthoracic and transesophageal, is the reference
method for morphological characterization of the TV and evaluation of TR
severity. Currently, to evaluate valvular morphology, advanced echocardiographic
methods are used, such as three-dimensional echocardiography which allow, through
the anatomical visualization of the tricuspid complex components and the
relationship with the surrounding structures, for the identification of the TR
etiology and the definition of the most appropriate therapeutic strategy. Through
echocardiography it is possible to quantify the valvular regurgitation, thanks to
a series of semi-quantitative and quantitative parameters. The severity of TR has
been classically defined by the presence of a vena contracta width
| Variable | Mild | Moderate | Severe | Massive | Torrential |
| VC (biplane) | 3–6.9 mm | 7–13 mm | 14–20 mm | ||
| EROA (PISA) | 20–39 mm |
40–59 mm |
60–79 mm |
||
| 3D VCA or quantitative EROA |
75–94 mm |
95–114 mm |
|||
| VC, vena contracta; EROA, effective regurgitant orifice area; 3D VCA,
three-dimensional vena contracta area; PISA, proximal isovelocity surface area. | |||||
In selected cases and only at the procedural stage, intracardiac echocardiography can play a complementary role for the correct display of structures that need to be properly evaluated during the implantation of specific transcatheter TV systems [68].
Computed tomography (CT) is not an integral part of the routine evaluation of TR. Nonetheless it becomes essential in patients undergoing TV surgery or certain transcatheter TV interventions, such as patients undergoing direct suture or tricuspid ring annuloplasty, as well as those undergoing TTVR, either orthotopic and heterotopic. It allows for adequate assessment of the TA position and size, the evaluation of structures surrounding the TV, such as the right coronary artery (RCA), at potential damage risk in some interventions. In addition, it is important to predict optimal fluoroscopic planes. Also, CT allows the measurement of IVC and SVC at different levels, as well as the evaluation of the relationships of the large veins with surrounding structures, which are key information in the preoperative phase of some interventions, as in the case of caval valve implantation (CAVI) [69].
Cardiac magnetic resonance imaging is currently the reference method for the quantization of volumes, and for the evaluation of RV morphology and function, and becomes essential if doubts persist after performing other non-invasive methods.
Right heart catheterization is scarcely used to evaluate patients with TR. So far its role has been relegated to reference method to exclude severe PH or to differentiate PH subtypes, especially if a surgical or transcatheter intervention is needed.
The role of coronary angiography is limited to cases in which the relationship between the RCA and TA need to be assessed, usually during annuloplasty systems deployment. At the beginning of annuloplasty procedure a guidewire is placed in the RCA via radial access, and at the end of the TV intervention or before if necessary, RCA angiography is performed to assess eventual iatrogenic coronary damage [70].
The gold standard for the treatment of severe TR still remains surgical repair, which according to American Heart Association/American College of Cardiology guidelines [71], is recommended in the following cases:
• in patients with severe TR at the time of left-sided valve surgery; • in symptomatic patients with primary severe TR; • in symptomatic patients with FTR poorly responsive to optimal medical therapy,
in which annular dilation coexists, in absence of severe PH or left-sided
disease; • in patients with progressive TR at the time of left-sided valve surgery if they
have suffered from right heart failure or in case of coexistent annular dilation
• in symptomatic patients with severe TR, who underwent prior left-sided valve
surgery, in absence of severe PH or RV systolic dysfunction; • in asymptomatic patients with severe primary TR and progressive RV dilation or
systolic dysfunction.
As for mitral valve, tricuspid surgical repair, when possible, should be
preferred to TV replacement [71]. It has been observed that when done at the
same time of left-sided surgery, concomitant tricuspid surgical repair has shown
to not impose additional operative risk and to be associated with RV reverse
remodeling and improvement of functional status [72]. The choice of a specific
surgical technique depends on the stage of natural history in which TR is
approached. In the presence of TA dilation without significant tethering
(coaptation height
Nevertheless, a large proportion of patients potentially candidate to TV surgery are rejected by the surgeon because of prohibitive perioperative risk, either because of referral in an advanced stage of the disease and also because often referral for redo-surgery [80].
With the aim of treating more patients, including those rejected by the surgeon, a great interest has emerged over recent years in the interventional cardiology community for developing minimally invasive transcatheter procedures to treat the “forgotten valve”. The complex anatomy of the valve, the important structures surrounding the tricuspid complex (conduction system, RCA, coronary sinus) at potential risk of damage during procedures, the absence of calcium in the large TA, the angle between the SVC/IVC and the TA, and the prominent subvalvular apparatus are just some of the challenges faced by the interventional cardiologist and underlines why many procedures have been thus far described as an “attempt” to approach the TV percutaneously. Current available transcatheter TV interventions can be divided into these two large groups:
TTVr systems, which include coaptation devices, whose objective is to increase
native leaflet coaptation or to occupy the regurgitant orifice area, and
annuloplasty systems, whose aim is to reduce the TA size. TTVR with heterotopic or orthotopic valves.
The main devices currently available, their potential advantages and disadvantages, and ongoing studies are presented in Tables 3,4,5.
| Device name | Manufacturer | Mechanism | Evidence | Advantages | Potential disadvantages | Image |
| MitraClip/ TriClip | Abbott Vascular (Santa Clara, CA, USA) | Edge-to-edge technique | • Case reports, case series • TriValve Registry [4] • TRILUMI-NATE [85] |
• Extensive interventional experience • Widely available • CE-mark approved technique • Preoperative CT not required |
• Limited steerability of the wire catheter in the right atrium (Mitraclip) • High quality TEE needed • Risk for entanglement with subvalvular structures (choardae tendinae) and PM leads • Challenging leaflet grasping (frequent large coaptation gaps needing multiple attempts) • Risk of SLDA (mostly with NT and XT device) |
 |
| PASCAL | Edwards Lifesciences (Irvine, CA, USA) | Edge-to-edge technique | • Case reports • Fam et al. [86] • CLASP TR [87] |
• Presence of a central spacer with major span width and less tension on the tricuspid leaflets • Clasps of the device operate independently (better leaflet insertion into the device and better span larger coaptation gaps) • Ability to reposition and recapture device if needed • Movement to some extent with the tricuspid leaflets (less leaflet tears and SLDA) • CE-mark approved technique • Preoperative CT not required |
• Local availability • Learning curve • Limited experience thus far |

|
| Mistral | Mitralix Ltd (Yokneam, Israel) | Edge-to-edge technique | • Case reports • Planer et al. [88] |
• Easy to implant (grasping of chordae of two adjacent leaflets to improve coaptation) • Possible to implant multiple devices in case of suboptimal result • Preoperative CT not required |
• Limited experience thus far |

|
| FORMA | Edwards Lifesciences (Irvine, CA, USA) | Coaptation device | • Perlman et al. [89] • Kodali et al. [90] |
• Easy to implant • Fully retrievable • Subclavian access • Preoperative CT not required |
• Voluminous device not addressing the pathophysiological alterations of FTR • Impact in RV pacing lead insertion • Risk of RV perforation |

|
| Trialign | Mitralign Inc. (Tewksbury, MA, USA) | Direct suture | • Case reports • SCOUT trial [91] |
• Previous surgical experience • Small device footprint (majority of native anatomy undisturbed for subsequent procedures) |
• Risk of leaflet and right coronary artery damage • Risk of mid-term device failure |

|
| TriCinch | 4Tech Cardio (Galway, Ireland) | Direct suture | • Case reports • PREVENT trial [92] |
• Previous surgical experience • Intraprocedural tension modification under echocardiography to optimize septolateral TV diameter |
• Risk of leaflet and right coronary artery damage • Risk of pericardial bleeding due to epicardial anchor • Risk of mid-term failure (device detachment) • Inferior vena cava dilation |

|
| Minimally invasive annuloplasty | Micro Interventional Devices (Newtown, PA, USA) | Direct suture | • Case report [93] | • Previous surgical experience • Sutureless implantation |
• Limited experience thus far |

|
| PASTA | NA | Direct suture | • Case report [96] | • Previous surgical experience • Improved pledget attachment (“double-bite” technique) |
• Risk of mid-term failure (device detachment) • Risk of leaflet and right coronary artery damage • Risk of annular dehiscence • Challenging technique. • Limited experience thus far |
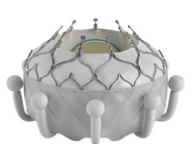
|
| Cardioband | Edwards Lifesciences (Irvine, CA, USA) | Direct ring annuloplasty | • Case-reports • TRI-REPAIR [98] • TR-EFS [99] • TriBAND [100] • Krober et al. [70] |
• Previous surgical experience • Experience with the device for mitral regurgitation treatment • CE-mark approved technique |
• Risk of right coronary artery damage and disengagement of anchors • High quality TEE needed • Heavily calcified TA unsuitable • Prolonged procedural time |

|
| CT, computed tomography; FTR, functional tricuspid regurgitation; NA, not available; PM, pacemaker; RV, right ventricle; SLDA, single leaflet device attachment; TA, tricuspid annulus; TEE, transesophageal echocardiography; TR, tricuspid regurgitation; TV, tricuspid valve. | ||||||
| Device name | Manufacturer | Mechanism | Evidence | Advantages | Potential disadvantages | Image |
| Caval Sapien Valve | Edward Lifesciences (Irvine, CA, USA) | CAVI | • Case reports • TRI-CAVAL [102] • Lauten et al. [106] |
• Extensive experience with TAVR • TEE guiding not mandatory |
• Risk for valve dislocation • Need for IVC and SVC landing zone preparation • Risk for hepatic vein occlusion with covered portion of valve • SVC often not suitable for balloon-expandable valve implantation (tapered dilation, PM/ICD leads) • Fails to address underlying TR pathology despite providing symptomatic relief via reduction in systemic venous congestion and caval backflow • Long-term impact of atrial ventricularization and progressive remodeling secondary to increased overload • Life-long anticoagulation often required |

|
| TricValve | P&F Products Features Vertriebs GmbH (Vienna, Austria) | CAVI | • Case reports [103] • Lauten et al. [106] |
• Easy to implant • Pre-stenting of the vena cava not required • Little radial force • Dedicated CAVI valve • Minimal risk for hepatic vein occlusion |
• Limited experience thus far • Fails to address underlying TR pathology despite providing symptomatic relief via reduction in systemic venous congestion and caval backflow • Long-term impact of atrial ventricularization and progressive remodeling secondary to increased overload • Life-long anticoagulation often required |

|
| Tricento | New Valve Technology (Hechingen, Germany) | CAVI | • Case reports [104,105] | • Easy to implant • Pre-stenting of the vena cava not required • Minimal risk for hepatic vein occlusion • Custom made based on pre-procedure imaging |
• Limited experience thus far • Logistical hurdles associated with patient-specific manufacturing • Fails to address underlying TR pathology despite providing symptomatic relief via reduction in systemic venous congestion and caval backflow • Long term impact of atrial ventricularization and progressive remodeling secondary to increased overload • Life-long anticoagulation often required |

|
| Navigate biopros-thesis | NaviGate Cardiac Structures Inc. (Lake Forest, CA, USA) | Orthotopic TV repla-cement | • Case reports [107] • Hahn et al. [108] |
• Abolishment of TR • Low profile (low risk of RVOT obstruction) • Valve replacement allows for more anatomies and etiologies to be treated • Promising feasibility and safety data thus far • Potential for valve-in-valve TTVR |
• Vascular access route (42-Fr transjugular) or transatrial with mini-thoracotomy • Bulky delivery system • Risk for damage to conduction system and/or ventricular septum • Afterload mismatch and worsening of RV failure from immediate TR resolution • Life-long anticoagulation often required |

|
| LUX-Valve | Ningbo Jenscare Biotechnology Co. (Ningbo, China) | Orthotopic TV repla-cement | • Case reports • Lu et al. [109] • Sun et al. [110] |
• Special radial force-independent anchoring mechanism to secure position • Abolishment of TR • Valve replacement allows for more anatomies and etiologies to be treated • Self-adaptive skirt to minimize paravalvular leak • Potential for valve-in-valve TTVR |
• Limited experience thus far • Only available for transatrial implantation with a mini-thoracotomy • Afterload mismatch and worsening of RV failure from immediate TR resolution • Life-long anticoagulation often required |

|
| EVO-QUE | Edwards Lifesciences (Irvine, CA, USA) | Orthotopic TV repla-cement | • Case reports • Fam et al. [111] • Preliminary data from TRI-SCEND [112] |
• 28-Fr transfemoral delivery system • Abolishment of TR • Catheter-based valve system familiar to operators • Multiplanar steerability allows for facilitated coaxial deployment in most anatomies • Valve replacement allows for more anatomies and etiologies to be treated • Potential for valve-in-valve TTVR |
• Risk for damage to conduction system • Afterload mismatch and worsening of RV failure from immediate TR resolution • Life-long anticoagulation often required |
|
| CAVI, caval valve implantation; Fr, French; ICD, implantable cardioverter defibrillator; IVC, inferior vena cava; PM, pacemaker; RV, right ventricle; RVOT, right ventricular outflow tract; SVC, superior vena cava; TAVR, transcatheter aortic valve replacement; TEE, transesophageal echocardiography; TR, tricuspid regurgitation; TTVR, transcatheter tricuspid valve replacement; TV, tricuspid valve. | ||||||
| Trial name | Summary | Device | Intervention/treatment | Patients enrolled | Primary outcome(s) | Recruitment status | Estimated primary completion date | Estimated study completion date |
| TRILUMI-NATE Pivotal RCT (NCT03904147) | Demonstrate the safety and effectiveness of the TriClip device in improving clinical outcomes in symptomatic patients with severe TR who are at intermediate or greater estimated risk of mortality or morbidity with tricuspid valve surgery | TriClip | Tricuspid valve repair vs medical therapy | 700 | Recruiting | August 2022 | March 2027 | |
| bRIGHT EU (NCT04483089) | Confirm the safety and performance of the TriClip device in a contemporary real-world setting | TriClip | Transcatheter heart valve procedure | 200 | Recruiting | December 2021 | December 2026 | |
| TRI-FR (NCT04646811) | Demonstrate that on the Packer composite clinical endpoint the tricuspid valve percutaneous repair strategy with clip for the tricuspid valve is superior to best (optimized) medical treatment in symptomatic patients with at least severe secondary TR | TriClip | Tricuspid valve repair vs medical therapy | 300 | Recruiting | February 2022 | August 2025 | |
| Berlin Registry of | Evaluate patients after interventional therapy | TriClip, Cardioband | Interventional therapy | 100 | Recruiting | December 31, | December 31, | |
| Right Heart Interven- | of valvular diseases of the right heart | mainly of TR | 2023 | 2023 | ||||
| tion (NCT04570163) | ||||||||
| TriCLASP | Collect data on the safety and effectiveness of | Edwards PASCAL | Edwards PASCAL | 300 | Recruiting | March 15, 2023 | March 15, 2028 | |
| (NCT04614402) | the PASCAL system in improving TR, functional status and quality of life in a post-market setting | transcatheter valve repair system | ||||||
| CLASP TR II Pivotal RCT (NCT04097145) | Determine the safety and effectiveness of the Edwards PASCAL transcatheter repair system in patients with symptomatic severe TR, at an intermediate or greater risk of mortality with tricuspid valve surgery | Edwards PASCAL | Edwards PASCAL transcatheter valve repair system vs optimal medical therapy | 825 | Recruiting | August 2022 | March 2027 | |
| MATTERS Study (NCT04071652) | Evaluate the acute safety of the implanted Mistral device post procedure and at 30 day follow up period | Mitralix Mistral device | Mistral Implantation | 20 | Recruiting | February 2023 | February 2025 | |
| Evaluate the long-term safety of the device. Demonstrate effectiveness of the Mistral device in reducing TR | ||||||||
| MATTERS II (NCT04073979) | Evaluate the acute safety of the implanted Mistral device post procedure and at 30 day follow | Mitralix Mistral device | Mistral Implantation | 10 | Recruiting | December 31, 2021 | December 31, 2023 | |
| up period. Evaluate the long-term safety of the device. Demonstrate effectiveness of the Mistral device in reducing TR | ||||||||
| SPACER (NCT02787408) | Assess the safety and the device performance of the Edwards tricuspid transcatheter repair system in patients with clinically significant, symptomatic TR who are at high surgical risk for standard tricuspid repair/replacement | FORMA device | Edwards tricuspid transcatheter repair system | 75 | Active, not recruiting | January 30, 2018 | December 2020 | |
| SCOUT II (NCT03225612) | Assess the safety and performance of the Trialign system for the treatment of symptomatic chronic FTR in patients with a minimum of moderate TR | Trialign | Mitralign Percutaneous Tricuspid Valve Annuloplasty | 60 | Recruiting | June 2018 | November 2022 | |
| NCT03632967 | Generate feasibility, safety, and performance data for the 4 Tech TriCinch Coil System in Symptomatic patients suffering from moderate to severe FTR with annular dilation | TriCinch | TriCinch Coil System Implantation | 7 (15) |
Terminated | July 14, 2020 | July 14, 2020 | |
| NCT03294200 | Generate feasibility, safety, and performance data for the 4 Tech TriCinch Coil System in Symptomatic patients suffering from moderate to severe FTR with annular dilation | TriCinch | TriCinch Coil System Implantation | 18 (60) |
Terminated | July 14, 2020 | July 14, 2020 | |
| STTAR (NCT03692598) | Evaluate the safety and performance of the MIA device in patients with chronic FTR | Minimally invasive annuloplasty | Minimally invasive annuloplasty implantation | 60 | Unknown | July 2020 | July 2021 | |
| TriBAND (NCT03779490) | Evaluate the safety and the effectiveness of the Cardioband Tricuspid Reconstruction System | Edward Cardioband TR | Transcatheter Tricuspid valve repair | 150 | Recruiting | July 24, 2019 | June 30, 2027 | |
| TRICI-HF (NCT04634266) | Asses if TTVT is associate with a morbidity and mortality reduction | Percutaneous CE-mark approved techniques for TTVT: Tricuspid Cardioband, Triclip, PASCAL | Transcatheter tricuspid valve treatment plus optimal medical therapy | 360 | Not yet recruiting | June 1, 2021 | December 31, 2026 | |
| NCT03700918 | First-in human study | DaVingi TR system | DaVingi TR system | 15 | Recruiting | December 2021 | June 2022 | |
| HOVER (NCT02339974) | Determine the short-term safety ( |
Edwards Sapien XT | Heterotopic Implantation of the Edwards | 30 | Active, not recruiting | January 2021 | January 2022 | |
| implantation of the Edwards Sapien XT valve in the inferior vena cava for the treatment of severe TR in patients who are inoperable or at very high surgical risk for tricuspid valve replacement | Sapien XT transcatheter valve in the inferior vena cava | |||||||
| TRICUS | Investigate the safety and performance for at | TricValve system | TricValve System | 10 | Active, not | July 2021 | July 2026 | |
| (NCT03723239) | least 30 days and up to 5 years | implantation | recruiting | |||||
| TRICUS STUDY Euro | Investigate the safety and performance for at | TricValve system | TricValve System | 35 | Active, not | May 5, 2021 | March 2025 | |
| (NCT04141137) | least 30 days and up to 5 years | implantation | recruiting | |||||
| TRAVEL | Evaluate the safety and effectiveness of LUX- | LUX-Vale | Tricuspid valve | 150 | Recruiting | June 2026 | June 2026 | |
| (NCT04436653) | Valve transcatheter tricuspid valve and delivery system which are intended to use in symptomatic patients with severe TR and high surgical risk | replacement system | ||||||
| TRISCEND (NCT04221490) | Evaluate the safety and performance of the Edwards EVOQUE Tricuspid Valve Replacement System | Edwards EVOQUE system | Transcatheter tricuspid valve replacement | 200 | Recruiting | October 2025 | December 2025 | |
| TRISCEND II (NCT04482062) | Pivotal trial to evaluate the safety and effectiveness of the Edwards EVOQUE tricuspid valve replacement system | Edwards EVOQUE system | Edwards EVOQUE System in conjunction with OMT vs OMT | 775 | Recruiting | June 2024 | June 2028 | |
| NCT04100720 | Evaluate the safety and technical performance of the Cardiovalve transfemoral system for | Boston Cardiovalve system | Cardiovalve transfemoral tricuspid valve | 15 | Not yet recruiting | December 2023 | December 2028 | |
| tricuspid valve replacement | ||||||||
| NCT04433065 | Early feasibility study to gain early clinical insight into the performance of the Intrepid transcatheter tricuspid valve replacement system | Medtronic Plc Intrepid System | Intrepid TTVR System | 15 | Recruiting | October 31, 2021 | November 15, 2026 | |
| NCT04905017 | Early feasibility study to gain early clinical | Trisol system | Transcatheter tricus- | 15 | Not yet | December 2021 | July 2027 | |
| insight into Trisol system safety and perfor- | pid valve replacement | recruiting | ||||||
| mance to treat patients with moderate or | ||||||||
| greater TR | ||||||||
| KCCQ, Kansas City Cardiomyopathy questionnaire; MAE, major adverse event; MIA,
minimally invasive annuloplasty; 6MWT, 6-minutes walking test; NYHA, New York
Heart Association; OMT, optimal medical therapy; SADE, serious adverse device
effects; SAE, serious adverse events; TEE, transesophageal echocardiography; TR,
tricuspid regurgitation; TTVR, transcatheter tricuspid valve replacement; TTVT,
transcatheter tricuspid valve treatment; VAD, ventricular assist device. | ||||||||
The MitraClip system (Abbott Vascular, Santa Clara, CA, USA), as shown in the TriValve (International multisite transcatheter tricuspid valve therapies registry) registry [4], is the most used TTVr system so far, probably because of the local availability and acquired experience over years with the same device in the treatment of mitral regurgitation. The Mitra-Clip device is a cobalt-chromium polyester-covered system with 2 arms able to grasp 2 leaflets. Although the 24 French (Fr) delivery system can be advanced both transjugularly and transfemorally, the most used access is the latter one. Until recently there have been only two clip sizes available, the 4 mm (NTR) and 6 mm (XTR). The new fourth generation MitraClip G4 (NTR, XTR, NTW and XTW) has been recently made available. This new system allows independent leaflet capture, in order to reduce single leaflet device attachment (SLDA). Furthermore, NTW and XTW devices have 50% wider clip arms. Improved results with the MitraClip system on the TV have been obtained in patients with the following clinical and echocardiographic features [81, 82, 83]:
• primary TR with valve prolapse or secondary TR with normal leaflets; • central/anteroseptal TR jet; • coaptation depth • coaptation gap • leaflet length • absence of global RV dysfunction (longitudinal and circumferential RV
dysfunction, as assessed by RV strain).
The TriClip (Abbott Vascular, Santa Clara, CA, USA) device holds the same
clip-based technology as MitraClip, but consists of a new, steerable guiding
catheter system suitable for the right side of the heart, facilitating the
interventional cardiologist to properly grasp and clip the TV leaflets. As well
for the MitraClip system, the new generation TriClip G4 has been recently made
available. 30-day and 6-month outcomes of the TriClip system in patients with
The Pascal device (Edwards Lifesciences, Irvine, CA, USA) consists of a 10 mm
central nitinol woven spacer that acts as a filler in the regurgitant orifice and
is attached to the valve leaflets by two paddles and clasps. Independent leaflet
grasping is possible and device repositioning and recapture are feasible. After
the initial publication of a compassionate use study [86], the CLASP TR
(Edwards Pascal TrAnScatheter Valve RePair System in Tricuspid Regurgitation
Early Feasibility Study) trial showed that in 34 patients with a mean age of 76
and
The spiral-shaped Mistral device (Mitralix Ltd, Yokneam, Israel) was intended to
improve leaflet coaptation by grasping the subvalvular apparatus. The device is
advanced transfemorally using an 8.5 Fr delivery system and is carried up into
the right ventricle. At this point the spiral is anchored and tightened around
the chordae tendineae of 2 adjacent leaflets which then constitute a
unique structure. The first-in-human experience has been recently reported,
showing at least 1 TR grade reduction in all patients [88]. No MAE were observed
at 30-day follow up. The device showed a significant reduction of key TR
echocardiographic parameters, as PISA EROA (0.15 [interquartile range, IQR
0.14–0.21] cm
The Forma spacer device (Edwards Lifesciences, Irvine, CA, USA), available in three sizes (12, 15 and 18 mm), is a foam-filled polymer balloon that provides
surface for native leaflets to coapt, promoting an edge/body-to-spacer leaflet
coaptation. It is advanced with a steerable delivery catheter from left
subclavian vein through a rail anchored at the septal portion of the RV apex. The
rail is also anchored to the subclavian vein, as a sort of PM. Two studies
reported the first experience with the device [89, 90]. Kodali et al.
[90] reported an early feasibility study, in which 29 highly comorbid patients
with a mean age of 75.9 undergoing Forma spacer device implantation for
Other coaptation devices currently under investigation include: the CroiValve system (CroíValve, Dublin, Ireland) and the Cerclage-TR block (Tau PNU MEDICAL Co. Ltd., Busan, Korea).
The Trialign system (Mitralign, Inc., Tewksbury, MA, USA) attempts to
percutaneously mimic the modified Kay surgical technique (obliteration of the
posterior leaflet) [76]. The goal is to achieve a bicuspidalization of the TV via
transjugular route. Bicuspidalization is obtained by delivering 2 polyester
pledgets via the right ventricle through the TA. A radiofrequency wire is used to
cross the annulus at the appropriate depth (2 to 4 mm from the base of the
leaflet). Pledgets are fixed at the posteroseptal and anteroposterior commissures
(the second pledget is fixed 2.4–2.8 cm from the first one) and then cinched,
resulting in plication of the posterior leaflet. If RV dilation is greater than
could be effectively treated with a single-pledget annular device, it’s possible
to implant 2 pairs of pledgets to obtain a greater TA size reduction. RCA
angiography is performed following implantation to check for eventual RCA damage.
The SCOUT (Percutaneous Tricuspid Valve Annuloplasty System [PTVAS] for
Symptomatic Chronic Functional Tricuspid Regurgitation) trial [91], an early
feasibility study of the Trialign device, enrolled 15 patients with moderate to
severe TR, advanced NYHA functional class (
The TriCinch system (4TECH Inc., Galway, Ireland) consists of two components: a stainless-steel screw that is placed in the antero-posterior portion of the TA and a self-expandable nitinol stent, variable in size (27–43 mm). The two components are connected by a Dacron band. The delivery system is advanced via the transfemoral route. After screw implantation, RCA angiography is needed to rule out RCA damage. At this point the stent and the whole system is tensioned to remodel and reduce the TA dimension and TV area, until satisfactory outcomes are observed by echocardiography. Implantation is then completed by deployment of the self-expandable stent in the IVC. In a second-generation device the screw tip has been replaced by a nitinol coil anchor. Preliminary data of 24 patients treated with the first-generation device TriCinch Screw Tip in the PREVENT (Percutaneous Treatment of Tricuspid Valve Regurgitation with the TriCinch system) study have been made available. The device was successfully implanted in 18 patients with a reduction of at least one grade of TR in 94% of cases. A hemopericardium occurred in 2 patients, RCA damage in 1 patient, while 5 subjects had late annular detachment of the device. The prevalence of severe TR was reduced from 80% to 40% [92]. Six-month follow-up was available only for 4 patients without a mention regarding echocardiographic parameters.
The minimally invasive annuloplasty-tricuspid (MIA-T) device (Micro Interventional Devices, Inc., Newtown, PA, USA) consists of ultra-low-mass, polymeric, biocompatible PolyCor anchors that are tensioned by the thermoplastic MyoLast polymer. The system is designed to obliterate the posterior leaflet by implanting the anchors between the posterolateral and anteroposterior commissures, replicating a surgical annuloplasty procedure on the end of a catheter. The MIA-T delivery system is a 12 Fr catheter. The anchors firmly grip the myocardium of the beating heart, are compatible with wall-motion and prevent device withdrawal. The radiopaque markers allow for visibility of the anchors on imaging modalities and provide guidance for the deployment during the procedure [93]. A locking mechanism secures the bicuspidalization in place when satisfactory TR reduction is achieved as confirmed by imaging, after the implantation of the anchors. So far, data on 3 subjects surgically treated with MIA-T device are available [93]. Implantation was successfully performed in all 3 patients, with no procedure or device related adverse events, no anchor detachments and a moderate/severe to mild/trace TR grade reduction, which remained consistent at 6-month follow-up.
Pledget-assisted suture tricuspid annuloplasty (PASTA) is a “percutaneous
surgical” procedure aiming to create a double-orificed TV, similar to the Hetzer
double-orifice suture technique [94, 95]. Percutaneous transthoracic access via
the RV apex and the transjugular route are viable access sites. Greenbaum
et al. [96] recently reported the first-in-human PASTA implantation in
an 83 years-old man with torrential TR (vena contracta width 22.9 mm), severe TA
dilation (18.12 cm
The Cardioband system (Edwards Lifesciences, Irvine, CA, USA) is a direct ring annuloplasty device previously used in patients with functional mitral regurgitation [97]. The goal of this device is to improve TR via annular size reduction. The procedure is performed by placing a 24 Fr sheath in the femoral vein, but a guidewire needs to be inserted in the RCA via radial access both as a landmark for Cardioband implantation and precautionary in case coronary interventions become necessary due to vessel damage during the procedure. An adjustable, incomplete surgical-like Dacron ring is affixed along the atrial side of the TA using up to 17 anchors, starting at the antero-septal commissure, advancing from the anterior to the posterior annulus. Under real-time echocardiography and fluoroscopic guidance, the entire device is then cinched through the pre-mounted wire within the implant [98]. The TA length (from the anterior leaflet to the coronary sinus) assessed using CT is important to determine the size of the device (from 73–80 mm, total anchors required 12, to 113–120 mm, total anchors required 17). Careful exclusion of heavy calcified annuli or leaflets is also needed, as well as an appropriate evaluation of the landing zone distance to the RCA, because of possible deformation resulting from the reduction of the TA.
Six-month data of the TRI-REPAIR (TrIcuspid Regurgitation RePair with CaRdioband
Transcatheter System) trial have been recently published [99]. Thirty patients
with severe TR, TA diameter
Data presenting 30-day outcomes from an early feasibility study in the United
States have also been published [99]. The Cardioband device was successfully
implanted in 28/30 patients (93.3%). In 2 cases there were anchor disengagements
with no clinical consequence and no further worsening of TR. At 30 days there was
a significant reduction in the end-diastolic septolateral TA (45.2
Preliminary data of the ongoing TriBAND (Transcatheter Repair of Tricuspid
Regurgitation With Cardioband TR System Post-Market Clinical Follow-Up Study)
trial similarly showed favorable outcomes at discharge and 30 days in patients
with
Recently Korber et al. [70] reported first real-world data of 60
patients undergoing Cardioband implantation for
Other annuloplasty systems currently under investigation include: the Millipede tricuspid ring, the transatrial intrapericardial tricuspid annuloplasty (TRAIPTA) and the DaVingi TR system (Cardiac Implants LLC, Wilmington, DE, USA), for which a first-in-human study is ongoing.
Many pathological TV anatomies remain unsuitable for TTVr systems, leaving valve replacement as a viable alternative. TTVR can be orthotopic or heterotopic. Challenges related to the anatomy of the TV, TA, right atrium and venae cavae make both procedures complex.
CAVI with either balloon-expandable or self-expandable valves into the venae cavae (IVC or both SVC and IVC) is thought to reduce the backflow to the venae cavae, improving venous systemic congestion, but not eliminating TR itself.
The growing confidence in balloon-expandable bioprostheses implantation for the
treatment of severe aortic stenosis (Edwards Sapien 3/XT, Edwards Lifesciences,
Irvine, CA, USA) has influenced the initial off-label use of these devices for
severe TR treatment [101]. In advanced severe TR, the SVC frequently shows a
tapered dilatation, which is not suited for balloon-expandable valve
implantation. Moreover, backflow into the SVC is often lower as compared to that
into the IVC due to hydrostatic pressure. This explains how CAVI with
balloon-expandable valves have been mostly performed only into the IVC. The
cavo-atrial junction anatomy and wall compliance of venae cavae may preclude the
direct implantation of a balloon-expandable valve and require the preparation of
a landing zone to facilitate the fixing of the valve by implanting a
self-expanding stent into the vena cava. The TRICAVAL (Treatment of Severe
Secondary TRIcuspid Regurgitation in Patients With Advance Heart Failure With
CAval Vein Implantation of the Edwards Sapien XT VALve) study was a prospective,
randomized, open label trial directly comparing CAVI versus optimal medical
therapy in patients with severe TR, high surgical risk, and NYHA class
Limitations of off-label use of other valves and the complexity of venae cavae anatomy reiterated the need for dedicated CAVI valves. The TricValve device (P&F Products Features Vertriebs GmbH, Vienna, Austria) consists of two pericardial tissue self-expandable valves on a nitinol stent frame, one specifically for the IVC and the other for the SVC. The IVC valve was designed with the upper segment protruding into the right atrium and the biological valve located above the diaphragm to protect the abdominal vasculature from systolic backflow and avoid occlusion of hepatic veins. For the SVC valve, the stent frame has a funnel shaped with the upper and lower segments tapered to facilitate sufficient fixation at the cavo-atrial inflow. The proximal stent segment is mounted with a tri-leaflet bovine pericardial valve and a sleeve covering the inside down to the base of the leaflets to prevent paravalvular leakage. The valves have minimal radial forces and don’t require pre-stenting of the venae cavae. Lauten et al. [103] reported the first in-human experience with the TricValve system. After successful implantation there was an immediate reduction of caval backflow and at 12-month follow-up the patient experienced a significant improvement in NYHA functional class, hepatic synthetic function and 6MWD.
The Tricento CAVI device (New Valve Technology, Hechingen, Germany) consists of a bi-cavally anchored nitinol stent deployed top down from the SVC into IVC. A lateral bicuspid porcine pericardium valve, requiring only a low closing pressure, allows inflow into the right atrium. The device is inserted tranfemorally through a 24 Fr delivery system. Since there is a high inter-individual anatomical variability, the device needs to be custom made for each patient. The upper and lower stent diameter need to be oversized by about 20% to reduce the risk of endoleak and a variable length of non-covered struts extend proximally to provide stability while avoiding hepatic vein obstruction. Toggweiler et al. [104] described the first-in-human case of successful implantation into a 74-year-old woman. After this report, other cases have been reported [105].
Lauten et al. [106] published the first compassionate series of patients (N = 25) using 3 different devices (the balloon-expandable Sapien XT/3 valve in 18 patients and the self-expandable valves TricValve and Directflow in 6 and 1 patient, respectively). Procedural success was achieved in 23/25 (96%) patients, with single (only inferior) or bicaval implantation in 19 (76%) and 6 (24%) of them, respectively. Two valve migrations requiring surgical intervention were reported. Significant improvements in NYHA functional class were observed at 30-day follow-up.
A series of unresolved concerns have thus far prevented the spread of the
technique, mainly limiting its use as symptomatic therapy in severely compromised
patients with advanced TR. Common issues among interventional cardiologists are
represented by long-term hemodynamic consequences of the therapy (right atrium
ventricularization or progressive remodeling secondary to increased overload),
the clinical significance and the impact on arrhythmic burden and RV function of
leaving TR itself untreated, the feasibility of further PM implantation or
transcatheter interventions, as well as the optimal antithrombotic treatment. In
addition, anatomical restrictions should be considered. CAVI suitability includes
in fact the demonstration of pulsatile caval backflow and adequate venae cavae
measures (
The complex anatomy of the TV and several pathophysiological and structural alterations sustaining FTR already discussed make developing dedicated bioprostheses for native TV particularly challenging. First, the dimensions of the TA may require large transcatheter valves along with large venous access. Second, the implantation of the valve in a non-calcific TV may be a further issue. Third, the proximity of the conduction system to TA and the interaction with the prosthesis might result in permanent conduction disturbances, requiring PM implantation, which in turn might represent an important issue because prostheses themselves might potentially prevent the subsequent implantation of a RV lead. To date, predominantly 3 transcatheter bioprostheses have been implanted in humans:
• the NaviGate stented valve, • the LUX-Valve, and, • the EVOQUE system.
Other prostheses in their initial phase of evaluation are the CardioValve (Boston Medical, Shrewsbury, MA, USA), Trisol (Trisol Medical, Yokneam, Israel), Intrepid (Medtronic Plc, Minneapolis, MN, USA) TRiCares (TRiCares SAS, Paris, France) and VDYNE (Jean Boulle Medtech, Luxembourg, Belgium) valves, for some of which an early feasibility study is ongoing.
The self-expanding tricuspid NaviGate bioprosthesis (NaviGate Cardiac Structures Inc, Lake Forest, CA, USA), available in 5 sizes (36, 40, 44, 48, and 52 mm), is an atrioventricular valved stent with a tri-leaflet design, which includes: patented partially dehydrated tissue technology, a self-expandable nitinol stent with atrial winglets, radially arranged ventricular graspers for anchoring and special fabric to promote fibrous ingrowth of biological material to aid in inter-chamber sealing of the device. The nitinol stent is wider in the ventricular region, giving it a classical conic configuration that reduces the transvalvular gradient and minimizes the RV outflow tract obstruction (low profile prosthesis, 21 mm, with little material protruding towards the ventricle). The valve is introduced through a 42 Fr delivery catheter via trans-jugular access. Alternatively, a trans-atrial approach after minimally invasive thoracotomy can be performed. There is an ongoing effort to develop a transfemoral delivery system. After the first orthotopic use of GATE TV stent in a native TV has been described [107], the early outcomes from the first 30 patients treated with this device has recently been reported [108]. In an elderly population with a median age of 75, severe TR and multiple comorbidities, undergoing compassionate GATE TTVR, device success was achieved in 26 patients (87%). Device implantation was performed via transatrial approach in 25 patients (83%) and via transjugular access in 5 patients (17%). Technical failure occurred in 4 patients, 2 of which were implanted transjugularly and 2 transatrially, with the latter needing conversion to open surgery. In-hospital mortality occurred in 3 patients. At 30 day-follow up 74% of patients had none to mild TR. At median follow up of 127 days there was only 1 further mortality. Of patients alive at follow-up, 62% were in NYHA functional class I or II, with no late device-related adverse events
The self-expanding bovine LUX-Valve system (Ningbo Jenscare
Biotechnology Co, Ningbo, China) is a radial force–independent orthotopic TTVR
device. It consists of 4 components: a tri-leaflet prosthetic valve with treated
bovine pericardium; a self-expandable nitinol stent including an atrial disc; a
bird tongue-shaped interventricular septal anchor and 2 expanded
polytetrafluoroethylene-covered graspers for leaflet fixation. The stent
bioprosthesis is available in 4 sizes (30 to 55 mm) and 8 skirt-shape disc models
intended for TA diameters of 25 to 50 mm. The LUX-Valve can be delivered via a 32
Fr catheter through a minimally invasive right thoracotomy and transatrial
approach under TEE and fluoroscopy guidance. The first-in-human experience was
recently published [109]. Twelve patients with severe TR, NYHA functional class
The EVOQUE system (Edwards Lifesciences, Irvine, CA, USA) consists of a
tri-leaflet bovine pericardial tissue valve, a nitinol frame, and a fabric skirt.
The outer diameter is variable: 44 mm, 48 mm, or 52 mm. The system is designed to
replace the TV in a native TA via a transfemoral 28 Fr percutaneous approach. The
prosthesis has a unique valve design allowing leaflets, chords, and annulus
engagement to achieve secure placement. Atraumatic anchors respecting native
anatomy and compatible with pre-existing leads are part of the prosthesis
structure. Recently, the first-in-human compassionate use of transfemoral TTVR
with the EVOQUE system in 25 patients was published [111], and preliminary data
of the TRISCEND (Edwards Transcatheter Tricuspid Valve Replacement: Investigation
of Safety and Clinical Efficacy Using a Novel Device) trial were presented [112].
In an elderly population of 56 patients with moderate TR (
Patients to be considered by the Heart Team for TTVr or TTVR should include
those with isolated severe TR, persistent symptoms despite optimal medical
therapy, and which have been deemed unsuitable for surgery. Exclusion of severe
LV dysfunction (LV ejection fraction
The largest prosthesis available in active clinical trials is the 52 mm EVOQUE
system. While larger valves could soon be tested in clinical trials, subjects
with a severe TA dilation should be considered either for TTVr other than
edge-to-edge technique (as a spacer device) or CAVI device. The latter in
particular requires for the patient to be suitable the pulsatile caval backflow
demonstration and adequate venae cavae measures (
Last but not least, important clinical considerations should be made. Procedural time is particularly long with some procedures, such as for Cardioband implantation. Thus, the risks and benefits should be adequately assessed in patients with impaired renal function, to avoid acute kidney injury in a high risk background of increased systemic venous pressure. A leitmotiv of studies published thus far on TTVr and TTVR is the risk of bleeding events, often the main determinants of MAE, so before considering TTVR, and subsequently lifelong anticoagulation, bleeding risk should be accurately assessed. Furthermore, local availability and expertise affect the choice of the device that can be offered to the patient.
We can further simplify the strategy for treatment of TR based on anatomical and pathophysiological factors. First, we can assess the coaptation gap size and severity of apical leaflet tethering by TEE. For all patients with coaptation gap size lower than 10 mm we should then assess apical leaflet tethering. If no apical leaflet tethering is detected, we can pursue by performing CT to assess TA for the evaluation of annuloplasty device. If deemed appropriate we can consider the latter, if not, we can consider edge-to-edge repair or even replacement. For mild to moderate apical leaflet tethering, with less than 10 mm coaptation gap we can consider straight away either edge-to-edge repair or replacement. If instead severe apical leaflet tethering exists, we should move on to assess RV and TA size by CT, as should be done for a coaptation gap above 10 mm. If severe RV or right atrial enlargement exist, specifically of the anulus, rendering inappropriate TTVR, then we should consider a spacer or CAVI device. On the other side, if right ventricle and annular size are deemed appropriate for TTVR systems, then we should reassess with transthoracic echocardiography, or better with TEE or cardiac magnetic resonance, to further proceed according to severity of RV dysfunction. If significant RV dysfunction is present, we can consider a spacer or CAVI device once again. On the other side, in absence of severe RV dysfunction we can consider TV replacement.
Despite often being “forgotten”, TR remains a common and important valvular heart disease. Significant TR is no longer thought as a marker of disease but is now widely seen as an important contributor of cardiac morbidity and mortality, independently of age, LV and RV function, atrial fibrillation and PH. Knowledge of anatomical details of the TV, together with the differences in the pathophysiology of FTR is paramount when approaching patients with TR. The complex anatomy of the TV and the heterogeneous pathophysiology of TR explain myriads of attempts to approach percutaneously the TV. Several TTVr and TTVR have been showed efficacy and safety when used on the TV. In the near future, these percutaneous approaches could provide an effective treatment option for more not suitable for surgery.
BARC, bleeding academic research consortium; BMI, body mass index; CAVI, caval valve implantation; CI, confidence interval; CT, computed tomography; EROA, effective regurgitant orifice area; FTR, functional tricuspid regurgitation; HR, hazard ratio; ICD, implantable cardioverter defibrillator; IQR, interquartile range; IVC, inferior vena cava; KCCQ, Kansas city cardiomyopathy questionnaire; LV, left ventricular; MIA-T, minimally invasive annuloplasty-tricuspid; MLHFQ, Minnesota living with heart failure questionnaire; MVARC, Mitral valve academic research consortium; NYHA, New York Heart Association; PASTA, pledget-assisted suture tricuspid annuloplasty; PH, pulmonary hypertension; PISA, proximal isovelocity surface area; PM, pacemaker; RCA, right coronary artery; RV, right ventricular; SAVR, surgical aortic valve replacement; SLDA, single leaflet device attachment; SVC, superior vena cava; TA, tricuspid annulus; TAPSE, tricuspid annular plane systolic excursion; TAVR, transcatheter aortic valve replacement; TEE, transesophageal echocardiography; TR, tricuspid regurgitation; TRAIPTA, transatrial intrapericardial tricuspid annuloplasty; TTVr, transcatheter tricuspid valve repair; TTVR, transcatheter tricuspid valve replacement; TV, tricuspid valve; VO2, volume of oxygen.
FC, MG wrote the paper. GGS critically revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
