1 GCA-Centro Spoke AO Cannizzaro, Catania, IRCCS Centro Neurolesi Bonino-Pulejo, 98124 Messina, Italy
2 Stroke Unit, AOU Policlinico Universitario, 98125 Messina, Italy
3 Neurorehabilitation Unit, IRCCS Centro Neurolesi “Bonino Pulejo”, 98124 Messina, Italy
Academic Editor: Parisa Gazerani
Abstract
Chronic migraine belongs to the “chronic long-duration headaches”, and it is associated to high burden and significant economic impact. Treatment for both episodic (EM) and chronic migraine (CM) is based on the management of acute attacks and their prevention. For moderate/severe attacks, pharmacological therapies are triptans, dihydroergotamine nasal sprays or injections or neuroleptics, non-steroidal anti-inflammatory drugs, and corticosteroids. Chronic migraine belongs to the “chronic long-duration headaches”, and it is associated to high burden and significant economic impact. Treatment for both episodic (EM) and chronic migraine (CM) is based on the management of acute attacks and their prevention. For moderate/severe attacks, pharmacological therapies are triptans, dihydroergotamine nasal sprays or injections or neuroleptics, non-steroidal anti-inflammatory drugs, and corticosteroids. The pathophysiology of CM is characterized by an abnormal activation of the trigemino-vascular system in the meninges causing a neurogenic inflammation, which explains the use of anti-inflammatory during attacks. It seems that the objective of the preventive therapy with the botulin toxin OnaBoNT-A consists in interrupting the release of CGRP and other neuropeptides as well as the activation of C-fiber nociceptor and of the nearby A-delta fibers. The protocol for migraine treatment with OnaBoNT-A injections consists of 31–39 pericranial injection sites involving seven muscle groups bilaterally in specific areas of the head and neck, with a total dose of between 155 and 195 units, every three months. The severe adverse events reported with high doses of botulin toxin for spasticity, have not been reported for CM treated with OnabotA at the labeled dose. The established improvement with onabotulinumtoxinA treatment in CM patients had a positive impact not only in reduction monthly headache days but also in improving quality of life, with reduction in both healthcare resource utilisation (HRU) and work impairment. Aim of this review was to give an overview on the use of BoNT-A in patients with CM, giving practical advices on the clinical indications.
Keywords
- chronic migraine
- onabotulinum toxin A
- botulin toxin
- calcitonin gene-related peptide (CGRP) pathway
Migraine is one of most disabling chronic pathology [1] with a negative impact on patients’ quality of life. The disease affects approximately 2% of the general population [2], and the prevalence is about 7% in men and around 24% in women. Moreover, it also affects about 4% of children [3, 4]. The World Health Organization Global Burden of Disease Study 2010 showed that migraine was the fourth most disabling medical disorder among women [5], and the seventh most disabling medical disorder overall worldwide.
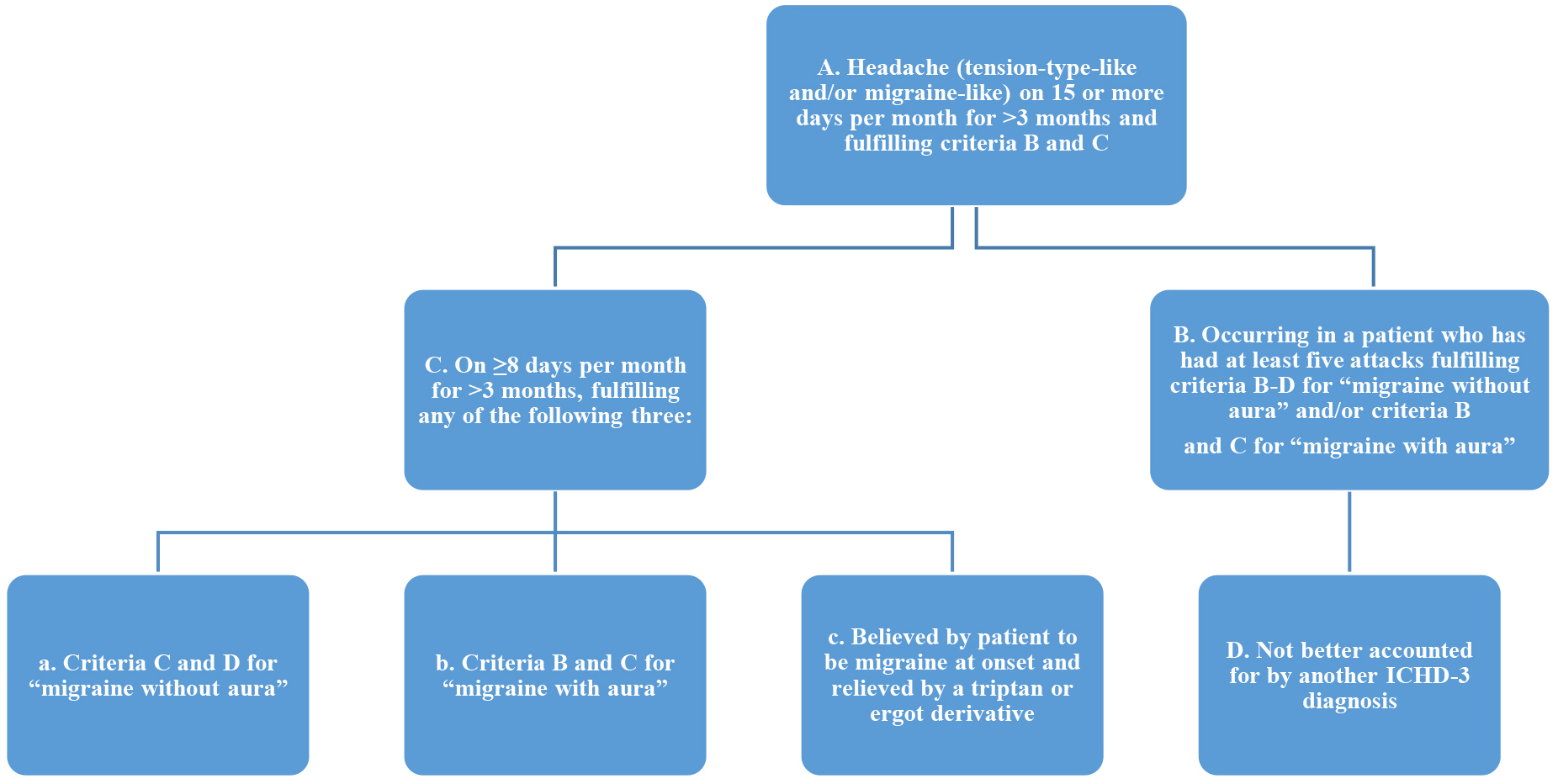
Migraine can be classified as with or without aura (characterized by visual, sensory or language symptoms) and as chronic or episodic based on the duration of symptoms. Chronic migraine (CM) is defined by the International Headache Society (IHS) as “headache on at least 15 days a month, for more than 3 months, and 8 of these headache days must be migraine headaches, or relieved by a triptan or ergot derivative and no medication overuse (Fig. 1) [6].
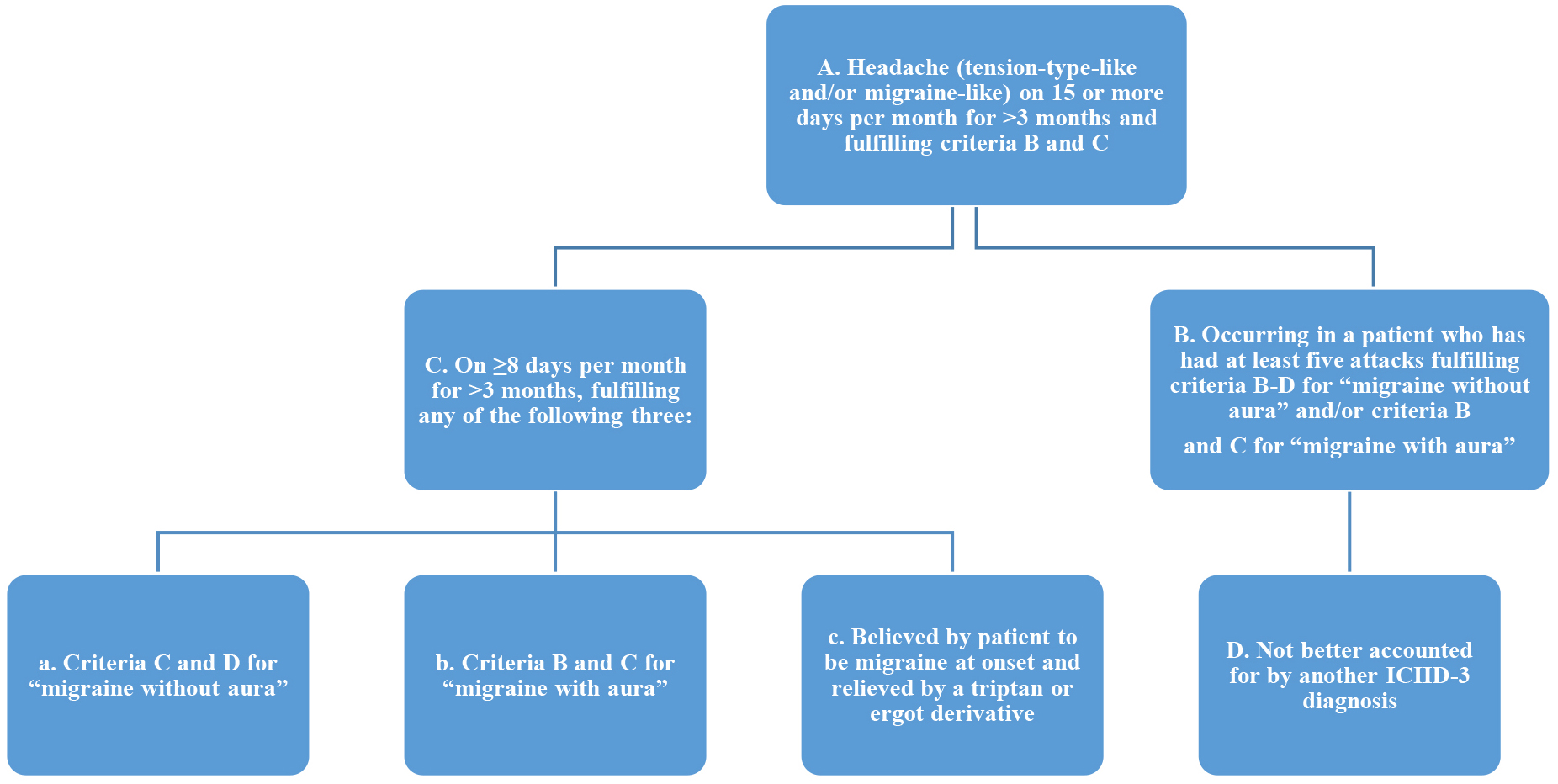 Fig. 1.
Fig. 1.ICHD-3 Diagnostic Criteria for CM. *Legend: CM, chronic migraine; ICHD-3, International Classification of Headache Disorders (third revision).
Chronic migraine, belongs to the “chronic long-duration headaches” and it is associated to high burden and significant economic impact [7]. It has been reported that CM patients lost at least 5 days of household work over a 3-month period compared with 24% of those with EM [7]. Treatment for both episodic and chronic migraine is based on the management of acute attacks and their prevention. Mild and sporadic migraine attacks can be managed with non-pharmacologic interventions, such as laying down in dark room, cold compresses or pressure on the painful areas, and avoiding migraine triggers. For moderate/severe attacks, pharmacological therapy is needed. Triptans should be taken early during the attack, with pain reduction usually within 2 hours, and should not be used more than two days per week in order to decrease the risk of rebound headaches. Other drugs commonly taken are the dihydroergotamine nasal sprays or injections or neuroleptics, non-steroidal anti-inflammatory drugs and corticosteroids. However, the treatments usually are slight efficient and limited by side-effects, as unintentional abuse [8]. On the other hand, although more than 40 drug therapies are used to prevent migraine, by now, only five agents are approved by the US Food and Drug Administration (FDA) for the prevention of EM: the beta-adrenergic blocking agents, propranolol and timolol, and the anticonvulsants divalproex sodium or sodium valproate and topiramate [9].
Furthermore, the only therapies approved for the prevention of CM are the onabotulinum toxin A (BoNT-A) [10], and the monoclonal antibodies that target the calcitonin gene-related peptide (CGRP) pathway [11].
Aim of this paper was to give an overview of the treatment with BoNT-A for CM, giving useful clinical indications. This may offer a guide on the use of botulinum toxin for the preventive treatment of migraine, including the sites and dose of injection, the approved indications for usage, the reported adverse events and impact on social problem.
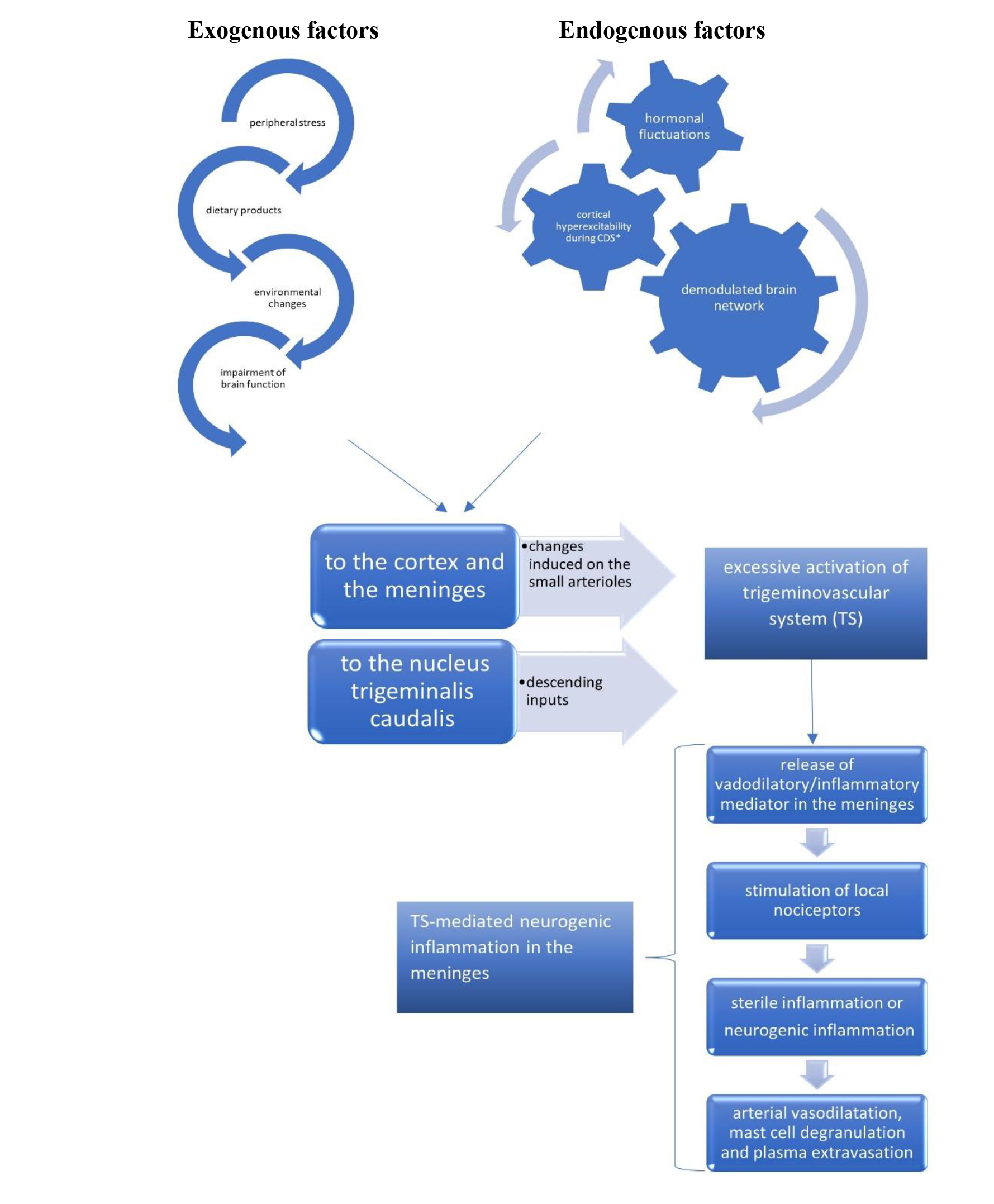
Significant progress has been made in the understanding of migraine pathophysiology, which is fundamental for the implementation of treatment options. Migraine is considered a primary central nervous system disorder involving several pathways, including the meningeal blood vessel [1] flow and the activation of some cortical and subcortical structures [12]. The pathophysiology of migraine is still debated [12]. The beginning of a migraine attack might be related to the activation of C-fiber nociceptors in the dura by the diffusion to the meninges of the neuropeptide calcitonin gene–related peptide (CGRP) and neurotrasmitters from the cerebral cortex during the spreading depression [13, 14]. The C-fiber nociceptors and A-delta pain fibers [15] and, subsequently, the second order sensory neurons of the trigeminocervical complex in the brainstem are activated [11]. Triggers are exogenous factors (as peripheral stress, dietary products, environmental changes) [12], that activate sensory afferents, as well as endogenous factors (spreading depression, hormonal fluctuation, demodulated brain networks) (Fig. 2, Ref. [12, 16, 17, 18, 19]) [20].
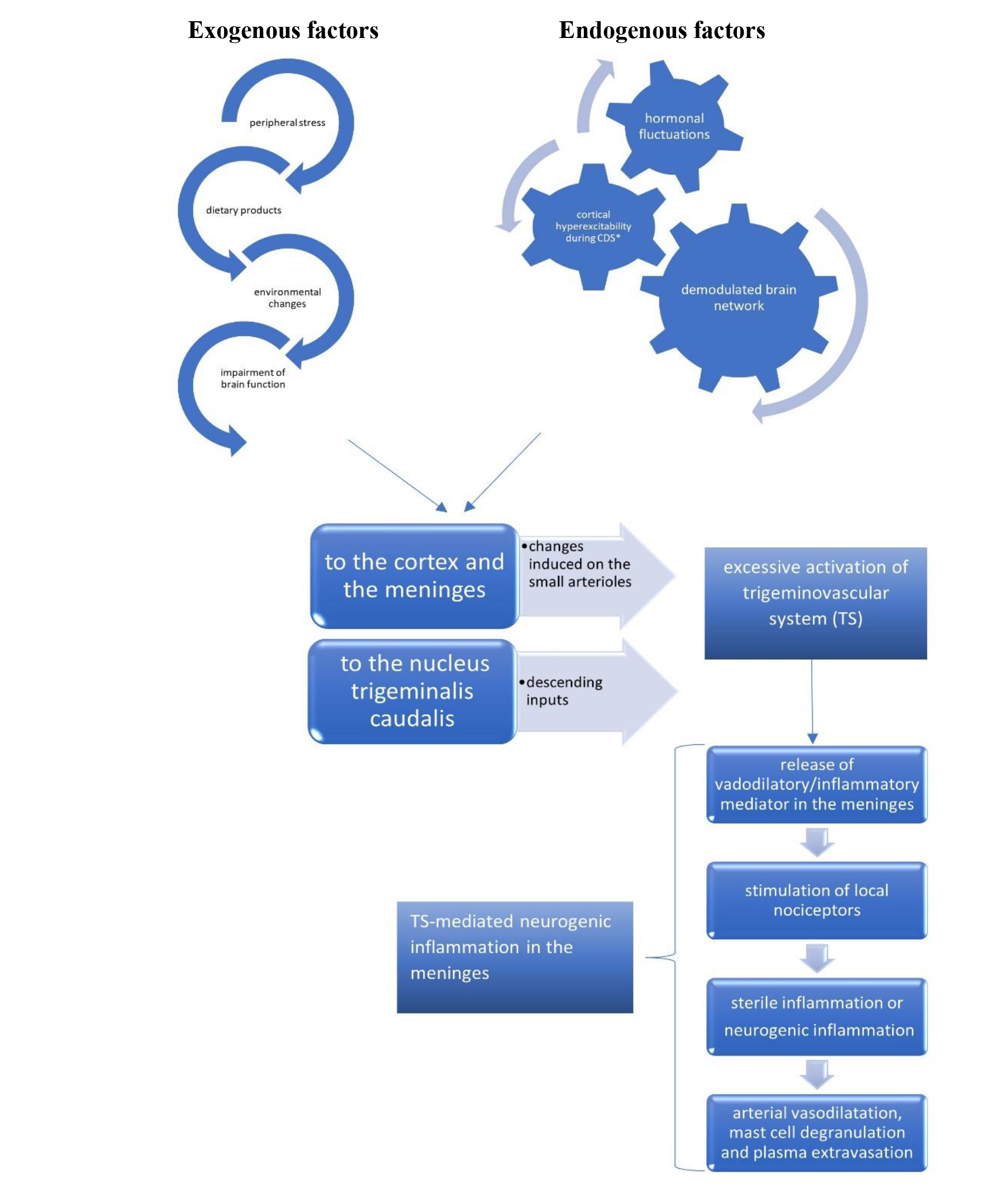 Fig. 2.
Fig. 2.The pathophysiological mechanisms involved in migraine attacks, with the pivotal role of the trigeminovascular system. CDS, cortical depression spreading.
Therefore, the pathophysiology is characterized by abnormal activation of the trigemino-vascular system in the meninges causing a neurogenic inflammation, that explains the use of anti-inflammatory during attacks [16]. The neuroinflammation lead to a double response: (i) a peripheral sensitization, mediated by an excessive response of primary afferent nociceptive neurons to external exogenous stressful [21], expressed phenotypically by throbbing pain, exacerbated by bending over or coughing [17]. The nociceptors and inflammatory mediators involved are bradykinin, histamine, serotonin, prostaglandin E2 and interleukins 1, 6 and 8 and tumour necrosis factor-alpha [16, 22], and (ii) a central sensitization which is caused by hyperexcitability of nociceptive neurons in the dorsal horn of the spinal cord [16]. This process is mediated by the CGRP, which has a pivotal role in [11] the nociceptive transmission and migraine chronification.
It seems that the objective of the preventive therapy with OnaBoNT-A consists in interrupting the release of CGRP and other neuropeptides as well as the activation of C-fiber nociceptor and of the nearby A-delta fibers [15].
OnaBoNT-A consists of two polypeptide chains joined by a disulfide bond. There are seven serotypes of botulin toxin: from A to G. However, the form that has been approved for the treatment of migraine is the onabotulinum toxin A, OnaBoNT-A [9, 23]. Moreover, the toxin has been utilised to treat craniocervical dystonia, spasticity, detrusor instability, hyperhidrosis and hypersalivation. The oral ingestion of OnaBoNT-A causes intoxication and produces acute and life-threatening muscle paralysis known as botulism [24]. Indeed, the BoNTs prevents the pre-synaptic release of the neurotransmitter acetylcholine at the synaptic level of neuromuscular junction causing dose-dependent, reversible flaccid paralysis [25, 26]. At the pre-synaptic space there are vesicles, nearby the terminal nerve [27], creating a pool filled by neurotransmitters, whose contents could be released in the synaptic junction to activate a signal transmission process after exocytosis. This last process is realized only after assemblation of syntaxin 1A and SNAP-25 (synaptosomal-associated protein), on the plasma membrane and synaptobrevin 2 (VAMP2) on the vesicular membrane to bridge the two opposite membranes in the ‘fusion’ step [20, 28]. The fusion complex VAMP/synaptobrevin/SNAP-25 and syntaxin, is called SNARE complex [29]. When OnaBoNT-A is injected, it is endocytosed and enters in the cell cytoplasm cleaving the C-terminus of SNAP-25, target of botulinum neurotoxins types A (BoNT-A) and BoNT-E proteolytic mechanism both in motor nerve terminals and in sensory nerve terminals [30]. As a result, the peripheral release of neuropeptides and inflammatory peptides [25] is blocked as well as the release of CGRP [28], distributed in the peripheral and central nervous system (CNS) [31] playing a therapeutic role in migraine [32].
CGRP is released in the meninges and in the nucleus trigeminalis caudalis. It is potentiated by the P2Y purinergic receptors in satellite glia cells [33], leading to cytokine release under the influence of sexual hormones, such as the 17-beta-estradiol. On the other hand, it is attenuated by the P2X3 receptor-mediated peripheral pain signal transduction in neuronal afferents [34]. The reduction in nociceptive activity in extracranial nerves or through reduction in blood CGRP levels in patients with chronic migraine has been showed by decrease in headache frequency [35].
Moreover, certain receptors, such as the transient receptor potential cation channel subfamily V member 1, TRPV1 [36], play an important role in pain transmission and sensitization, and could be targeted by other drugs.
The first case of botulism has been published in 1820, and the pathogen agent was successively isolated in 1897 and named as bacillus botulinum and then as clostridium botulinum [37]. Taking into consideration its potential in the medical field, botulin toxin has been used in 1977 for the treatment of strabism, and in the 1989 was also approved for blepharospasm and hemifacial spasm [37]. Only in the nineties, OnaBoNT-A has been used for tension-type headache, and, since then, several case reports with positive effect of chronic migraine were reported [9, 10, 11]. The first clinical trial was described in 2000 by Allergan, that obtained the rights to Oculinum [38]. Even though the trial showed contrasting results with benefit only with low doses (25 UI) instead of higher doses (75 UI), an increasing interest on clinical trials has amplified the number of clinical trials on OnaBoNT-A in migraine. At first the OnaBoNT-A was approved only for preventive treatment of chronic migraine. Later two larger sample OnaBoNT-A vs placebo RDBPC trials of patients with chronic headache were carried out and showed the efficacy of OnaBoNT-A on reducing headache days per month more than 50% (32.7 vs. 15.0%, p = 0.027) [39]. Nevertheless, a later episodic migraine trial in 2007 showed no differences between OnaBoNT-A and placebo [25]. However, a post-hoc analysis showed positive results in patients with a headache frequency more than 12 days per month [23].
The next important trial was the phase III Research Evaluating Migraine
Prophylaxis Therapy (PREEMPT) involving 1384 patients [10]. Although the primary
endpoint of reduction in mean change in headache episodes from baseline failed, a
significant reduction in headache day per month as secondary endpoint has been
shown. However, the study demonstrated a slight difference in reduction of
headache days compared with placebo (2.3 days) and global pain intensity in
patients with CM. On the other hand, no significant findings for botulinum toxin
injection in the of tension type and episodic migraine were found [40, 41, 42]. The
efficacy of OnaBoNT-A in CM was particularly shown in the two randomised
placebo-controlled phase 3 clinical trials, PREEMPT I and II [10, 23]. Both
studies showed a decrease in headache days and migraine days with OnaBoNT-A as
compared to placebo [10, 23]. A pooled analysis of the two previous studies showed
a decrease from baseline in frequency of headache days by 2 days per month at
week 24 and at all other time points and a statistically significant improvement
(p
Moreover, in the large long-term open-label COMPEL (Chronic Migraine Onabotulinum toxin-A Prolonged Efficacy Open Label) study, Blumenfeld et al. [44] showed no differences in the number of moderate or severe headache days per month between patients with and without daily headache. However, a significant reduction in frequency of headache days at weeks 24, 60 and 108 was shown. The HIT-6 scores were significantly reduced at each time point in both groups from baseline (65.6 and 64.6, respectively) to 60 or less. For these reasons, OnaBoNT-A was approved only for CM [44].
A recent metanalysis showed that OnaBoNT-A, topiramate, and acupuncture reduce headache days and migraine days a week 12 with no significant difference between the three treatments in most of the outcomes investigated; however, topiramate caused higher adverse events and number of dropout. The authors concluded that, even though all of the three treatments were effective in the prophylactic treatment of CM, OnaBoNT-A should be considered as the first-choice respect to topiramate, because of the better effect in reducing headache days and migraine days a week 16 and the lower acceptability of topiramate, and the acupuncture, because of the several sessions needed (at least 10 sessions, one per week) [45] with important economic costs [46].
Moreover, a systematic metanalysis on the treatment of CM with OnaBoNT-A demonstrated its effectiveness compared to placebo, as well as the non-inferiority to the comparators valproic acid, topiramate and histamine, with a better safety and tolerability profile than oral prophylactics [47].
Suitable patients for treatment with BoNT-A toxin are (i) subjects over 18 years
old; (ii)
Because of the origin of migraine pain come from the meninges and the forehead, bridge of the nose, the temples, the back of the head, and neck, injections of peripheral scalp results in retrograde transportation through the connections (skull sutures) to the meninges, Gasserian trigeminal ganglion and occipital nerves through dorsal roots to the cervical spine [49, 50, 51].
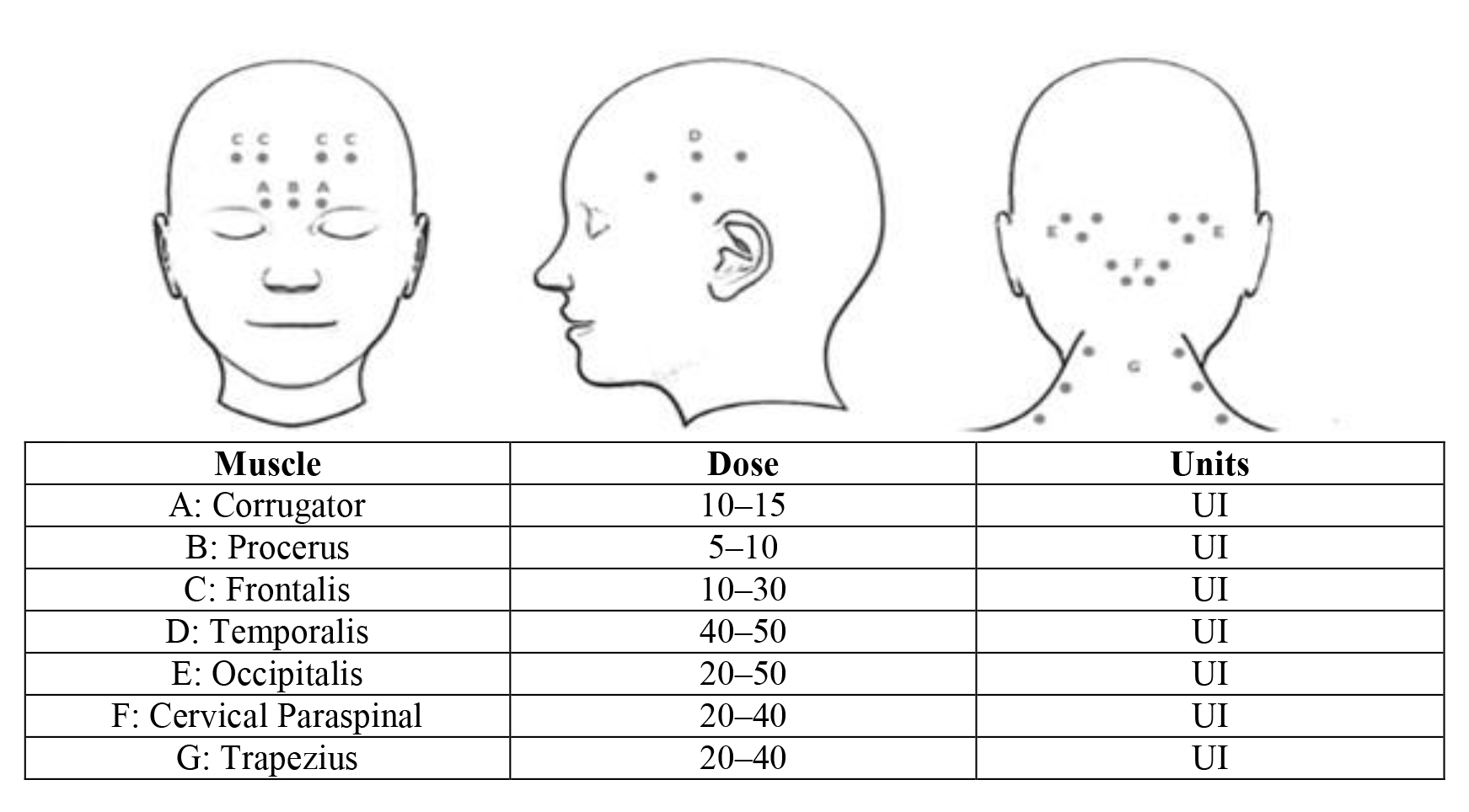
Therefore, the protocol for migraine treatment with OnaBoNT-A injections
involving of 31–39 pericranial injection sites consisting of seven muscle groups
bilaterally in specific areas of the head and neck (Fig. 3) [10, 23] with a total dose of between 155 and 195 units [44], every three months. About the
muscle groups, in the temporal region the injection should be done
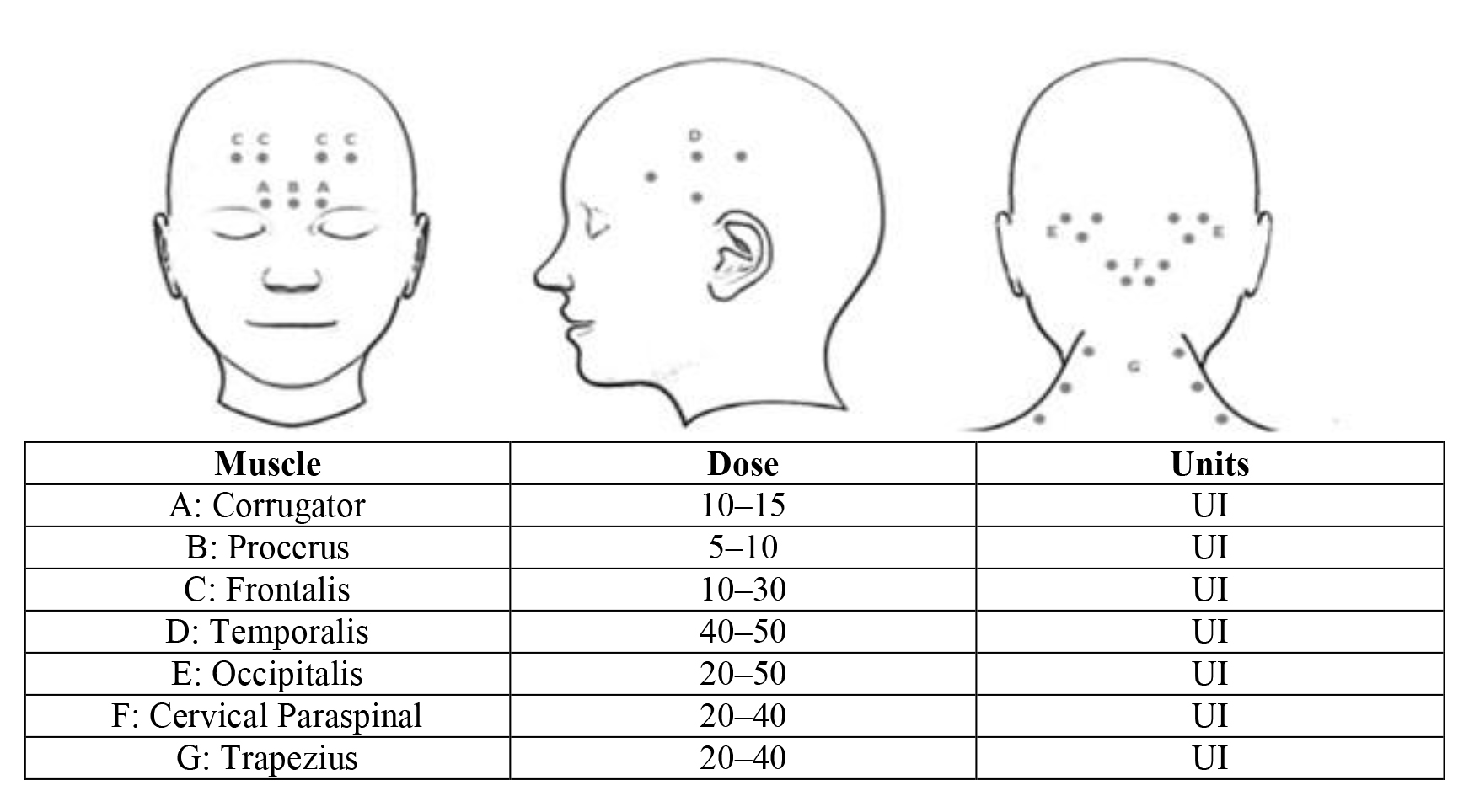 Fig. 3.
Fig. 3.The main injection sites for botulin toxin in the prevention of chronic migraine.
Recently, an algorithm of OnaBoNT-A in migraine has been proposed based on the
clinical response, evaluated by headache diaries or validated tools as: rating
scale to rate the intensity of headache [55], MIDAS or HIT-6 score for disability
[56, 57], MSQ for QoL [58], PGCI for patient’s impression of efficacy [59]. Four
categories have been classified: excellent responders, when the patients
experienced a reduction in headache days
The second group are called good responders for a reduction of headache days or
scores from
When a response from
Lastly, patients who respond with a reduction of headache days
| Responders | Headache days | 1 dose | 1 year treatment | 2 year | 3 year | ||
| Excellent | reduction |
started with a dose of 155 | Every 3 months (dose 155–195UI) | Continue monotherapy | Continue monotherapy | Every 4 months | Discontinuation |
| Good | reduction from |
started with a dose of 155 | Every 3 months (dose 195UI) | At 6 months consider OnaBont-A+ oral therapy | Continue politherapy | Every 4 months | Increase interval |
| Low | reduction from |
started with a dose of 155 | Every 3 months (dose 195UI) | At 3 months consider ONABONT-A+ oral therapy | At 9 months consider to switch to mAB | Every 3 months | OnaBOnt-A monotherapy every 3 months |
| Non- responders | reduction from |
started with a dose of 155 | Every 3 months (dose 195UI) | At 3 months ONABONT-A+ oral therapy | At 6–9 months consider to switch to mAB | / | / |
About the dosage of OnaBont-A+, a recent prospective study compared the efficacy
of 195 U in 172 patients to 155 patients treated with 155 U of OnaBont-A+. The
authors showed a higher efficacy of 195 U in reducing the mean headache days
every three months of evaluation period (p
The long-lasting efficacy of OnaBoNt-A+ has been shown in a long-term study of 2015 on 132 patients with CM [62]. The study showed that botulinum toxin is effective after one year of treatment with injection every three months, with only 9.3% of patients dropped after one year for lost of efficacy, likely due to worsening of migraine or a development of antibodies. Meanwhile, no failure after three years of treatment were obtained. The treatment should not be delayed after three-four months and should not be stopped unless worsening of migraine frequency and pain. As adverse events, the frontal and temporal atrophy muscles was reported in two patients without pain. However, injections were continued every four months [62].
In a small study with 90 patients with CM, it has been shown that patients injected during the afternoon showed a lower average of total number of headache days during 3 months following injection than the morning-treated patients (p = 0.0007) [63].
OnabotulinumtoxinA is the only agent specifically approved for the prevention of CM in 2012 [64].
Recently four monoclonal anti-CGRP antibodies (mAbs-CGRP) have been developed as
a new therapeutic for CM: one against the CGRP-R (erenumab) and three against the
CGRP peptide or ligand (eptinezumab, fremanezumab and galcanezumab). Although,
OnaBont-A and mAbs-CGRP showed a good efficacy, safety profile and tolerability
studies on comparison efficacy are missing. CGRP/CGRP-R mAbs are recommended for
the treatment of episodic migraine (
Galcanezumab is injected at the doses of 120 mg or 240 mg per month with a significant decrease in the average number of migraine days, 2.1 and 1.9 days compared to the placebo group [68].
Non-invasive neuromodulation could be of help in some cases where toxins and other pharmacological treatments fail.
The data from literature indicates that Transcranial magnetic stimulation, TMS, can consistently reduce the number of migraine days per month, mitigate the intensity and duration of migraine attacks for several days after TMS application, up to one month, interrupt migraine attack quickly, and improve the quality of life. In addition, TMS is safe and well-tolerated, also considering that the majority of trials employed subthreshold stimulation intensities. Unfortunately, the non-invasive TMS devices are relatively expensive, even if the method seems to be cost-effective in some specific cases: it is cheaper than botulinum toxin-A for chronic migraine treatment, as well as cheaper than the whole, complex pharmacological management of chronic migraine) [69, 70].
Besides, transcranial direct current stimulation (tDCS) is a promising effective, preventive, and safe method for migraine treatment. Indeed, a recent systematic review has shown that tDCS applied on the primary motor cortex (M1), as well as on visual cortex VC could reduce the number of migraine days per month in patients with migraine [71].
Finally, combined approaches may lead to some additional benefit in CM [18, 19, 72, 73], and should be considered the future on chronic intractable migraine. Combination of CGRP mAbs and onaBoNtA treatment in CM patients has been recently studied in 257 patients with an effective additive or synergistic result and well tolerated effect. However, CGRP mAb were discontinued more frequently than onabotulinumtoxinA (23.3 vs. 3.3%), usually for lack of insurance reimbursement (42%) or a lack of effect (21%) [74].
A recent study has shown, for the first time ever, the therapeutic effect of mindfulness practices associated with left dorsolateral prefrontal cortex anodal tDCS in improving the level of full attention and analgesic benefits in this patient population [75, 76].
The long-term real-world PREEMPT trials showed that about 40–73% of patients reported one or more adverse event with OnaBoNt-A [44, 77] consisting in neck pain (4%), musculoskeletal pain (2%), migraine or worsening of migraine (1–8.7%) and blurred vision (1%) [78, 79]. However, in patients continuing the injections, the incidence rate reduced in subsequent treatment session, showing a satisfied effect over the slight adverse events [80, 81].
The severe adverse events reported with high doses of botulin toxin for spasticity, have not been reported for CM treated with onabotA at the labeled dose [82]. The safety was shown even in infants breast-fed by lactating women treated for CM [83]. About safety in pregnancy women, adverse events were similar to the general population [84].
The established improvement with onaboNTA treatment in CM patients had a positive impact not only in reduction monthly headache days but also in improving quality of life, with reduction in both healthcare resource utilisation (HRU) and work impairment [85, 86]. Compared with episodic migraine, patients with CM showed diminished quality of life [87], a higher level of disability [88, 89], higher healthcare utilisation and reduced productivity, developing in a higher economic burden [87]. CM also negatively influenced the overall health as well as marital, childcare, relationships, career/financial achievement and stability [90]. Moreover, CM is associated with substantial disability, healthcare resource utilisation (HRU), and economic burden [88, 89, 90, 91, 92, 93, 94].
Notably, data from the PREEMPT clinical trials, the evaluation of quality of life in the REPOSE study, and resource utilization estimates the International Burden of Migraine Study (IBMS) have revealed the cost-effective resource of the onaboNTA in UK [95].
The multi-center REPOSE study showed a statistically significant reduction in headache-related hospitalizations and healthcare professional visits before baseline; reduction from 41.7% at baseline to 13.5% at visit 8 of family doctor visits as well as a reduction from 61.7% to 5.2% of visits to a medical specialist, at the end of the 2-year observation period. Moreover, there was a reduction in percentage of 77% and 78% of patients absent from school or work as well as an increased performance at school or work over the 2-year period. Therefore, no patients reported headache-related disability at visit 8 [96].
In addition, the COMPEL study showed the efficacy of onabotulinumtoxinA treatment also in headache-related HCP visits, emergency room and urgent care visits, and diagnostic tests in adults with CM [44].
Another real-world, open-label study of CM patients showed a reduction of emergency department visits (55%), urgent care visits (59%) and hospitalizations (57%) as compared to the 6 months before initiating botulinum toxin treatment [97].
Chronic migraine impacts the quality of life of the patients and, therefore, to reduce the frequency and duration of headache attacks should be the first object of the treatment. To this end, Botulin toxin has been shown as a valuable treatment for the prevention of CM. In low or non-responder patients a combination therapy with oral drugs or monoclonal antibodies (Erenumab, fremanezumab, and galcanezumab) should be considered, even though few studies are available [7]. However, the management of CM remains difficult because of the necessity of a better understanding of the pathophysiology of CM and the effect of different combination of CM therapy, as well as the discovery of others therapeutic targets.
RSC designed the manuscript structure. LR, and GR discussed and wrote the content and exchanged ideas and suggestions throughout the writing process. DM and CC exchanged ideas and suggestions throughout the writing process. RSC edited and critically revised the paper and gave the final approval for the version to be published. All authors have read and agreed to the published version of the manuscript.
Not applicable.
Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest. RSC is serving as one of the Editorial Board members/Guest editors of this journal. We declare that RSC had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Dr. Parisa Gazerani.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.