†These authors contributed equally.
Academic Editor: Foteini Christidi
Background: Enlarged perivascular spaces (ePVS) and white matter
hyperintensities (WMHs) are recognised neuroimaging lesions for symptomatic
and/or occult cerebral small vessel disease (CSVD) that are linked with the
predisposition to cardiocerebrovascular risk and neurocognitive impairment. This
study aimed to determine the interrelation between the WMHs and ePVS,
neurocognition, and cardiocerebrovascular risk profiles in asymptomatic
working-aged adults at a single-center population-based cohort. Methods:
Fifty-four asymptomatic subjects (mean age: 39.6
Cerebral small vessel disease (CSVD) is a pathologic process involving the small
penetrating arteries and arterioles (up to 200
WMHs are commonly found in ageing brain as bright areas of small non-cavitated
high signal intensities on fluid attenuated inversion recovery (FLAIR) and
T2-weighted MRI sequences. In contrast, ePVS are defined as cerebrospinal fluids
(CSF) filled cavities (
It has been previously shown that executive dysfunction may correspond with diffused white matter lesion and CSVD manifestation (i.e., WMHs and ePVS) in elderly individuals [13], whilst the overall severity of subcortical lesions is known to link with the reduction in speed of information processing and executive performance in patients with subcortical ischemic vascular dementia [14]. In contrast, such global cognitive deficits are not seen in those with additional heterogenous vascular dementia (i.e., dementia with mixed or spectrum of syndrome and pathophysiology) [15]. In fact, a significant relationship between WMHs severity and processing speed in older adults (65–80 years of age) has been reported [16]. Multiple assessments tools have been developed to assess changes in cognitive performance in affected and asymptomatic individuals. To date, the Weschler Adults Intelligence Scale (WAIS) is one of the most widely used psychometric instrument and enables assessing neurocognitive function for the perceptual reasoning index (PRI), processing speed index (PSI) and working memory index (WMI). The PRI assesses the ability of the subjects to visualize, understand, hand-eye coordination and work with non-verbal information. The PSI measures the subjects’ attending to visual material, visual perception and organization, visual scanning, and hand-eye coordination while the WMI assesses the ability to memorize, to hold and manipulate information in short-term memory and attention [17, 18].
Furthermore, WMHs and ePVS are characterized and rated in terms of their proportion (i.e., volume, location, size, and number of lesions). Hence, various visual rating scales have been developed to assess WMHs, including the Fazekas scale [19] which provide an overall impression of the presence and proportion of the lesions in the whole brain. Apart from that, the current advancement in neuroimaging technology has enabled various specific algorithmic techniques and tools to be developed which automatically detect the presence and proportion (i.e., number, volume, and location) of WMHs and ePVS to provide more concise delineations of these lesions [20]. Therefore, this study aimed to find the interrelation between the MRI-finding of occult CSVD (i.e., WMHs and ePVS) in asymptomatic, economically active adults with their cardiocerebrovascular risk prediction score and neurocognitive performances (perceptual reasoning, working memory and processing speed).
This study was conducted at the Hospital Universiti Sains Malaysia (HUSM), a suburban tertiary referral centre for neurological disorders in the East Coast of Malaysia with a catchment population base of over 4 million people. This study was conducted according to the guidelines of Declaration of Helsinki and ethics approval was obtained from the Human Research Ethics Committee-Universiti Sains Malaysia (USM/JEPeM/15030096).
Recruitment of subject’s population was through a simple convenience random sampling (subjects who attended Family Medicine Clinic at HUSM for their general medical consults). Eligible subjects recruited were asymptomatic (i.e., no prior medical, no neurological disease symptoms, and without any past or current history of major psychiatric disorder and/or developmental disorders) individuals, working-aged 25–65 years living in the suburban east coast peninsular Malaysia. A total of 203 subjects was initially screened. Their demographic and clinical details were stratified according to cardiocerebrovascular risk calculation by QRISK version 3 (QRISK3).
The QRISK3 version 2018 (http://www.QRISK.org/index.php; University of
Nottingham and EMIS, UK) is an online based cardiocerebrovascular risk calculator
which was used to calculate the percentage of disease risk (for next
10-years—including myocardial infarct and/or stroke and/or transient ischemic
attack) for all subjects. The projected risks were arranged according to the
following criteria: 0 to 10% = no/low cardiocerebrovascular risk; 10.1 to 20% =
moderate risk; and
 Fig. 1.
Fig. 1.Study flowchart of subjects’ identification and inclusion/exclusion.
The subjects underwent a brain scanning using 32-channel head coil, 3T MRI
Philip® Achieva (Philips Medical Systems, Best, The Netherlands).
Three-dimensional T1-weighted images were acquired using magnetization-prepared
rapid gradient-echo (MPRAGE) sequence in sagittal planes with echo time
(TE)/repetition time (TR) = 3.5/7.6 ms; inversion time = 864 ms; field of view
(FOV) = 250
Topographically, WMHs were categorized into two, i.e., periventricular, and deep WMHs. In contrast, ePVS has been recognized to be in several locations such as basal ganglia (along the proximal part of lenticulostriate arteries, e.g., globus pallidus, caudate nucleus, and putamen), centrum semiovale, midbrain (i.e., brainstem), and hippocampus. Thus, in this study WMHs and ePVS were identified and differentiated according to their signal intensities on MRI sequences, size, location, shape, and border as previously defined [21, 22].
Based on the STandards for Reporting Vascular changes on nEuroimaging (STRIVE) method, WMHs featured as increase intensity or hyperintensity on T2-weighted image, and FLAIR (best identified) MRI, and hypointense (decrease intensity) on T1-weighted image. Meanwhile, ePVS had similar signal intensity with CSF, reduced FLAIR and T1-weighted signal, increased T2-weighted signal and iso-intense on diffusion-weighted imaging (DWI) and T2*-weighted gradient recalled echo (GRE) signal [2]. With their variable location, size, shape, and number, WMHs and ePVS are recognized CSVD manifestations in subcortical gray matter and mainly in white matter [23, 24, 25].
In this study, the brain MRI images were visualized using MeDINria software
version 2.2 (Inria, National Institute for Research in Digital Science and
Technology, France) [26] to manually detect the presence of WMHs of presumed
vascular origin and ePVS from T2, T1, and FLAIR images. Their severity was
evaluated using visual rating scale Fazekas scale (3-level severity score) upon
FLAIR images [19, 27]. Two experienced neuroradiologists who were blinded to all
clinical data rated WMHs (deep and periventricular) and ePVS independently. The
intra- and inter-rater agreements were assessed in random from 54 subjects with
interval of longer than 1 month between the first and second readings. The kappa
coefficients for the inter-rater agreements were
The images were further processed for white matter lesion segmentation. This
procedure used to automatically detect and evaluate WMHs and to study the number
and volume of lesion. To do this, legion growth algorithm (LGA) as implemented in
the lesion segmentation tools (LST)
(http://www.statistical-modelling.de/lst.html) were used for statistical
parametric mapping at
Several subcortical and cortical structures were selected based on the presence of ePVS in subjects with CSVD neuroimaging manifestation. Subcortical segmentation volumes pertaining to caudate, putamen, pallidum, hippocampus, cerebellar white matter, and brain stem were extracted from 3D MPRAGE images using FastSurfer [29], a deep learning-based neuroimaging pipeline. Unlike traditional neuroimage processing pipelines which involve time-intensive optimization processes, FastSurfer employs fully convolutional neural networks (F-CNNs) for quick and efficient whole brain segmentation, cortical surface reconstruction and implements spherical mapping using a spectral approach. Using GNU parallel [30], and on high performance computing resources at National Supercomputing Centre, NSCC, (1 Fusionopolis Way, #17-01 Connexis South, Singapore) [30], whole brain segmentation for 54 subjects was completed within 23 hours.
In this assessment, the WAIS-Fourth edition,WAIS-IV (Pearson/PsychCorp, USA) was used to assess the general cognitive and memory abilities. The PRI was utilized to measure non-verbal reasoning and perceptual organization through three subtests including block design, matrix reasoning, and visual puzzle. WMI was used to measures specifically, simultaneous, and sequential processing, attention, and concentration, through two subtests including digit span and letter number sequencing. Lastly, PSI was used to measure speed of mental and grapho-motor processing, through two subtests including coding and symbol search. Scores obtained from each subtest were used for group-wise comparison and multimodal correlation.
The collected data were analyzed using Statistical Package for Social Sciences
(SPSS) software version 26.0 (IBM Corp., Armonk, NY, USA). An alpha (
Table 1 shows the general characteristics of the study population, which
consisted of 54 subjects (Fig. 1) with a mean age of 39.6 years
| Variables | Total (N = 54) | WMHs | ePVS | |||||
| Present (n = 18) | Absent (n = 36) | p-value | Present (n = 26) | Absent (n = 28) | p-value | |||
| Age, yrs.* | 39.6 |
46.6 |
36.0 |
0.00 | 43.12 |
36.3 |
0.02 | |
| Gender, n (%) | 0.84 | 0.22 | ||||||
| Male | 17 (29.8) | 6 (33.3) | 11 (30.6) | 10 (38.5) | 7 (25) | |||
| Female | 37 (64.9) | 12 (66.7) | 25 (69.4) | 16 (61.5) | 21 (75) | |||
| Ethnicity, n (%) | 0.19 | 0.63 | ||||||
| Malay | 48 (88.9) | 18 (100) | 30 (83.3) | 24 (92.4) | 24 (85.7) | |||
| Chinese | 4 (7.4) | 0 (0) | 4 (11.1) | 1 (3.8) | 3 (10.7) | |||
| Others | 2 (3.7) | 0 (0) | 2 (5.6) | 1 (3.8) | 1 (3.6) | |||
| Education level, n (%) | 0.30 | 0.54 | ||||||
| Low |
5 (9.3) | 1 (5.6) | 4 (11.1) | 3 (11.5) | 2 (7.1) | |||
| Intermediate |
22 (40.7) | 10 (55.6) | 12 (33.3) | 12 (46.2) | 10 (35.7) | |||
| High |
27 (50.0) | 7 (38.8) | 20 (55.6) | 11 (42.3) | 16 (57.2) | |||
| Smoking, n (%) | 0.37 | 0.35 | ||||||
| Non-smoker | 48 (88.8) | 15 (83.3) | 33 (91.7) | 22 (84.6) | 26 (93) | |||
| Ex-smoker | 4 (7.4) | 2 (11.1) | 2 (5.6) | 3 (11.5) | 1 (3.5) | |||
| Light smoker | 1 (1.9) | 0 (0) | 1 (2.7) | 0 (0) | 1 (3.5) | |||
| Moderate smoker | 1 (1.9) | 1 (5.6) | 0 (0) | 1 (3.9) | 0 (0) | |||
| Heavy smoker | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | |||
| Type 2 Diabetes, n (%) | 0 (0) | 0 (0) | 0 (0) | - | 0 (0) | 0 (0) | - | |
| Family History |
13 (22.8) | 7 (38.8) | 6 (16.6) | 0.26 | 11 (42.3) | 2 (7.1) | 0.004 | |
| Atrial fibrillation, n (%) | 0 (0) | 0 (0) | 0 (0) | - | 0 (0) | 0 (0) | - | |
| Hyperlipidaemia, n (%) | 0 (0) | 0 (0) | 0 (0) | - | 0 (0) | 0 (0) | - | |
| Hypertension, n (%) | 9 (15.8) | 6 (33.3) | 3 (8.3) | 0.02 | 8 (30.7) | 1 (3.5) | 0.01 | |
| CHO to HDL ratio * | 3.88 |
3.62 |
4.01 |
0.20 | 3.97 |
3.80 |
0.52 | |
| SBP (mmHg) * | 128.9 |
141.3 |
122.8 |
0.00 | 134.3 |
123.9 |
0.01 | |
| BMI (kg/m |
24.7 |
25.8 |
24.2 |
0.20 | 26.4 |
23.2 |
0.00 | |
| QRISK3 Score (%) * | 2.81 |
6.07 |
1.20 |
0.00 | 4.5 |
1.3 |
0.01 | |
| WAIS-IV Indices * | ||||||||
| PRI | 102.2 |
100.8 |
103.0 |
0.57 | 101.7 |
102.7 |
0.78 | |
| WMI | 108.0 |
107.3 |
108.1 |
0.93 | 107.4 |
108.5 |
0.82 | |
| PSI | 102.0 |
98.6 |
103.7 |
0.21 | 98.7 |
105.1 |
0.09 | |
| Note: data values are presented as number of subjects (n), with percentage
(%) in parentheses; light smoker ( | ||||||||
Table 1 describes the characteristic of study subjects with and without WMHs,
whereby 18 (33%; mean age: 46.6
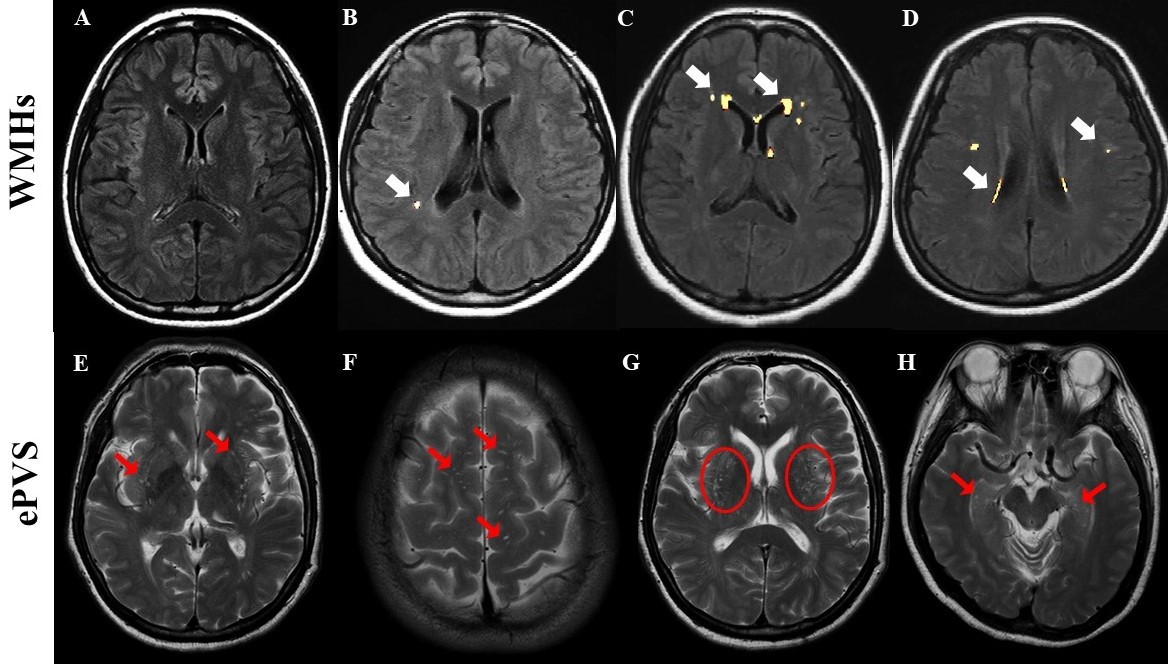 Fig. 2.
Fig. 2.Spatial distribution of white matter hyperintensities (WMHs) of presumed vascular origin and enlarged perivascular spaces (ePVS) among study subjects. Top rows represent spatial distribution of statistical probable voxels output from lesion segmentation tool (LST)-based lesion growth algorithm (LGA) (bright yellow highlighted) seen on fluid attenuated inverse recovery (FLAIR) images. (A) FLAIR image represent subject without WMHs (normal brain). (B) Represent subjects with deep WMHs (white arrows). (C,D) Represent subjects with both periventricular and deep WMHs (white arrows). Bottom rows represent the structural T2-weighted images of subjects with ePVS. (E,G) Represent subjects with multiple ePVS in basal ganglia (i.e., globus pallidus, caudate nucleus, and putamen). (F) Represent subjects with multiple ePVS in bi-hemispheric subcortical white matter such as centrum semiovale. (H) Represent subjects with multiple ePVS in bilateral hippocampus.
Table 1 also describes the characteristic of study subjects with and without
ePVS. A total of 26 subjects (48%) had ePVS, whereby all of them had
| Average Volume | Median (Interquartile Range, IQR) | p-value | |
| ePVS Present (n = 26) | ePVS Absent (n = 28) | ||
| Caudate Nucleus | 3483.3 (3305.0–3689.7) | 3564.9 (3356.7–3686.8) | 0.85 |
| Globus Pallidus | 1949.7 (1902.9–1939.7) | 2010.9 (1892.9–2045.3) | 0.71 |
| Putamen | 4891.8 (4829.5–5334.0) | 5281.0 (4920.6–5392.4) | 0.66 |
| Hippocampus | 3951.3 (3866.4–4109.3) | 4119.7 (3935.9–4303.6) | 0.22 |
| ePVS, enlarged perivascular spaces. | |||
Interestingly, 12 subjects (22%) had both WMHs and ePVS (i.e., combined lesion). Hence, among 18 subjects with WMHs, 6 subjects had WMHs alone (i.e., without ePVS). In contrast, among 26 subjects with ePVS, 14 subjects had ePVS alone (i.e., without WMHs).
The characteristic of subjects with and without WMHs and ePVS are listed in
Table 1. The association was corrected for multivariable non-colinearity. The
model fit assessment was perfomed using Hosmer-Lemeshow goodness-of-fit test,
fitted for age and QRISK3 (p
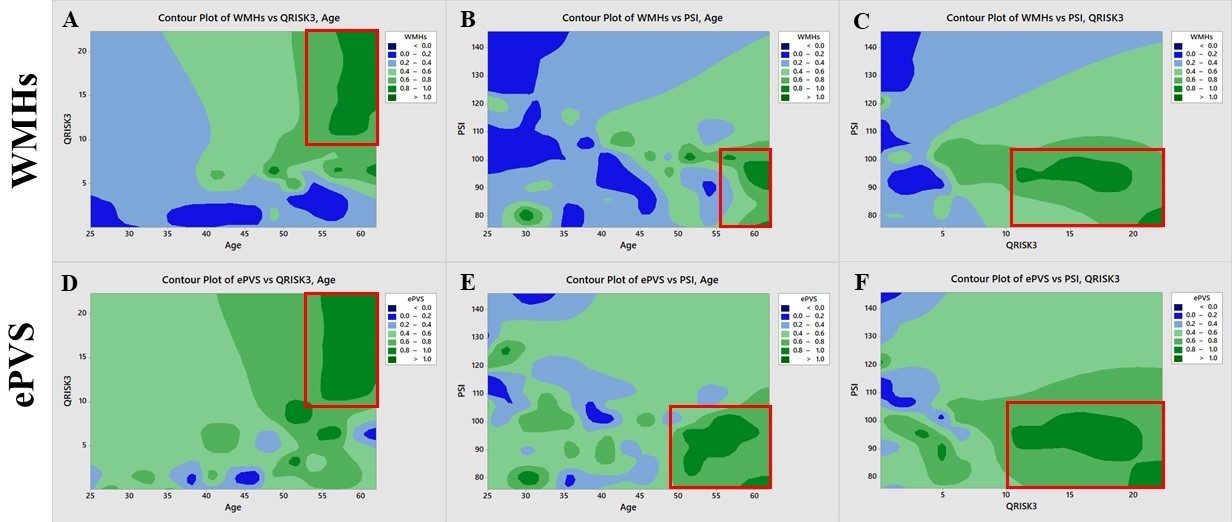 Fig. 3.
Fig. 3.Contour plots represent the graphical relationship between white
matter hyperintensities (WMHs), enlarged perivascular spaces (ePVS) with QRISK3,
age and processing speed index (PSI). (A,D) Relationship between (A) WMHs, (D)
ePVS with QRISK3, and age. Red box (with dark green shade) indicates increased
age had higher QRISK3 score (
| Risk Factors | WMHs Present (n = 18) versus absent (n = 36) | ePVS Present (n = 26) versus absent (n = 28) | ||
| Multiple Logistic Regression |
Multiple Logistic Regression | |||
| OR (95% CI) | p-value* | OR (95% CI) | p-value* | |
| Age |
0.91 (0.86 to 0.97) | 0.03 | 0.95 (0.89 to 0.99) | 0.03 |
| Family History |
2.07 (0.58 to 7.46) | 0.26 | 9.53 (1.86 to 48.91) | 0.01 |
| Hypertension | 5.50 (1.19 to 25.54) | 0.03 | 12.0 (1.38 to 104.34) | 0.02 |
| SBP |
0.89 (0.83 to 0.95) | 0.00 | 0.95 (0.91 to 0.99) | 0.02 |
| BMI | 0.92 (0.80 to 1.05) | 0.20 | 0.83 (0.72 to 0.96) | 0.01 |
| QRISK3 Score | 0.65 (0.48 to 0.88) | 0.00 | 0.75 (0.58 to 0.98) | 0.03 |
| WAIS-IV PRI | 1.01 (0.94 to 1.08) | 0.88 | 0.98 (0.92 to 1.05) | 0.53 |
| WAIS-IV WMI | 0.98 (0.93 to 1.03) | 0.40 | 0.99 (0.94 to 1.03) | 0.52 |
| WAIS-IV PSI | 1.04 (0.98 to 1.11) | 0.16 | 1.06 (1.00 to 1.13) | 0.04 |
| BMI, body mass index; CI, confidence interval; ePVS, enlarged perivascular
spaces; OR, odds ratio; PRI, perceptual reasoning index; PSI, processing speed
index; SBP, systolic blood pressure; WAIS, Weschler Adult Intelligence Scale
version IV; WMHs, white matter hyperintensities; WMI, working memory index;
| ||||
The presence of ePVS is also associated with age (OR, 0.95; 95% CI:
0.89 to 0.99), QRISK3 score (OR, 0.75; 95% CI: 0.58 to 0.98), and other
cardio-cerebrovascular disease risk factors (i.e., family history of
cardiocerebrovascular disease, hypertension, higher SBP, and higher body mass
index [BMI]). The prevalence of ePVS was found in much younger subjects compared
to WMHs, indicating that ePVS can appear in much younger age compared to WMHs.
However, WMHs was not associated with family history of cardiocerebrovascular
disease and BMI. We found that, gender, ethnicity, smoking status, and education
level were not associated with both WMHs and ePVS. Moreover, we found that WMHs,
specifically deep MWHs, was significantly associated with average volume of
globus pallidum (GP) but not with CN, putamen, hippocampus (standardized
In addition, subjects with both WMHs and ePVS (i.e., combined lesion) had higher
mean age (49.00
Based on Table 1, subjects with WMHs and ePVS had lower neurocognitive
performance (corrected co-variates, i.e., age, QRISK3 profiles), although the
difference was not statistically significant. However, we found that PSI had a
linear relationship with age (standardized
Additionally, in subjects with WMHs–the volume of WMHs had a linear association
with age and QRISK3 score, whereby the number of WMHs was associated with QRISK3
score (Table 4). The volume and number of WMHs was not associated with
cardiocerebrovascular risk factors (i.e., hypertension, SBP, family history, and
BMI) and subjects’ neurocognitive performances. On the other hand, subjects with
ePVS showed a reduced average volume of CN, GP, putamen, and hippocampus were
associated with increase age and QRISK3. Moreover, reduced average volume of
hippocampus also associated with a reduced subject working memory (WMI)
(standardized
| Variables | White Matter Hyperintensities (WMHs) | Enlarged Perivascular Spaces (ePVS) | ||||
| WMHs Volume | WMHs Number | Caudate Nucleus |
Globus Pallidus |
Putamen |
Hippocampus | |
| Age | 0.50** (−0.31*) | 0.52** (−0.08) | −0.50** (−1.03*) | −0.45** (−0.59*) | −0.24 (−0.80*) | −0.16 (−0.67*) |
| QRISK3 | 0.81** (0.93*) | 0.71* (0.71*) | −0.10 (0.818) | −0.16 (0.62*) | 0.04 (0.56*) | 0.13 (0.85*) |
| Hypertension | 0.49** (−0.00) | 0.41** (−0.05) | −0.07 (−0.21) | −0.42 (−0.09) | 0.10 (−0.72) | 0.12 (−0.19) |
| Systolic Blood Pressure | 0.61** (0.21) | 0.59** (0.21) | −0.16 (−0.13) | −0.11 (−0.13) | −0.05 (0.04) | 0.07 (−0.09) |
| Family History |
0.16 (−0.06) | 0.10 (−0.10) | 0.02 (–0.03) | 0.06 (0.04) | −0.11 (−0.08) | −0.16 (−0.23) |
| BMI | 0.07 (−0.13) | 0.16 (−0.04) | −0.01 (0.15) | 0.67 (0.25) | 0.45 (0.30*) | 0.19 (0.32*) |
| WAIS-IV PRI | −0.18 (0.04) | −0.10 (0.07) | 0.37** (0.33) | 0.28* (−0.02) | −0.18 (0.09) | 0.14 (−0.06) |
| WAIS-IV WMI | −0.21 (−0.01) | −0.10 (0.15) | 0.32* (0.07) | 0.28* (0.16) | 0.21 (0.04) | 0.30* (0.46*) |
| WAIS-IV PSI | −0.24 (−0.06) | −0.24 (−0.13) | 0.31* (−0.20) | 0.38** (0.05) | 0.13 (0.12) | 0.10 (−0.19) |
| Notes: r = Pearson correlation coefficient, | ||||||
We sought to determine the interrelations between the prevalence and proportions of WMHs and ePVS with the cardiocerebrovascular risk factors and neurocognitive profiles among asymptomatic individuals. Interestingly, out of 54 neurological asymptomatic and economically active middle-aged individuals, we found 33.3% and 48.1% with an incidental WMHs and ePVS from the MRI brain, respectively, with the severity ranging from 1 to 2, based on Fazekas scale. Moreover, ePVS was found mostly in subcortical white matter (i.e., centrum semiovale [92.3%]), basal ganglia including caudate nucleus (CN, 7.7%), globus pallidum (GP, 19.2%), and putamen (26.9%). Among subjects with ePVS, 30.7% had ePVS in hippocampus. Of note, 12 subjects had both WMHs and ePVS (combined lesion).
In this study, the presence, and the proportion of WMHs (irrespective of deep or periventricular) and ePVS was significantly associated with age, whereby asymptomatic older subjects had a higher prevalence. Our findings are in line with the previous studies, whereby older individuals had higher prevalence of WMHs and ePVS [32, 33, 34, 35, 36]. Moreover, multiple large-scale studies had supported that the increased prevalence of WMHs with aging, for example, Study of Health in Pomerania (SHIP) cohort study (N = 2367, age: 20–90 years) that reported the presence of WMHs (deep and periventricular) are explained by an increased age [37]. Additionally, several studies had also described the prevalence of CSVD (i.e., WMHs and ePVS) in much younger individuals for example; recent studies reported that, in subjects (age 18 to 50 years) with pre-existing (yet occult) CSVD manifestations had higher chances of developing ischemic stroke and vice versa [38, 39]. Moreover, Viana-Baptista and collegues also described young adults (mean age: 47.7 years) with cardio-cerebrovascular risk factors had higher prevalence of CSVD manifestation [40].
Besides, we also found the presence of WMHs and ePVS are associated with hypertension and SBP. These findings are supported by multiple studies that indicate cardio-cerebrovascular risk factors such as hypertension and elevated SBP affect small penetrating arteries of the brain leading to development of WMHs and ePVS [34, 41, 42, 43, 44, 45, 46].
In addition, Lai and colleagues supported that higher blood pressure was
associated with presence of WMHs hence suggested that intensive blood pressure
control aided in preventing the progression of WMHs [47]. Similarly, the Systolic
Blood Pressure Intervention Trial (SPRINT) study suggested that intensive control
(SBP
Moreover, in this study the cardiocerebrovascular risk was cumulatively measured as QRISK3 risk prediction score or percentage. Hence it is found that QRISK3 was associated with the presence and proportion of WMHs and ePVS. Whereby individuals with WMHs and ePVS (especially subject that had combined lesion) had higher QRISK3 score, hence had more prominent cardiocerebrovascular disease risk factors such as high SBP and hypertension despite being asymptomatic at the time of MRI brain scan. Besides, we also found significant association between higher QRISK3 score with aging. Recent study by Jung and colleagues in asymptomatic older population (N = 130, aged over 50 years), revealed that periventricular and deep WMHs was associated with age and cardiocerebrovascular risk factor (i.e., hypertension) [50]. Multiple studies had supported the finding that WMHs and ePVS are associated with aging and higher cardiocerebrovascular disease risk (i.e., hypertension and high SBP) and supported the notion that both of CSVD manifestation are particularly common in the world’s population including Caucasian [51], Afro-American and Afro-Caribbean [51, 52], and Asian populations such as Malays and Chinese Singaporeans [53, 54], Chinese populations [55], and Japanese populations [56]. Alarmingly, the prevalence of both CSVD manifestations have been reported to be higher in Asian population compared to western world [57].
The pathomechanism of WMHs and ePVS is still a subject in active research. However, it is well accepted that the pathological changes related to CSVD, and its manifestation are closely linked to the disrupted cerebral microcirculation [33] and characterized by occlusion of cerebral small vessel by age-related microvascular pathology (i.e., arteriolar tortuosity and venous collagenosis, age-related demyelination and loss of glial cells [41, 58, 59, 60]. Alongside several cardiocerebrovascular risk factors such as hypertension, obesity, and type-2 diabetes which in turn lead to decreased cerebral blood flow (cBF) and blood brain barrier (BBB) disruption in CSVD patients [61, 62].
In addition, the disturbance of interstitial fluid (ISF) drainage (i.e., due to waste product accumulation alongside the cerebral perforating arteries) and disrupted CSF-ISF exchange in glymphatic system (i.e., glympopathy) has been associated with the pathomechanism of WMHs and ePVS especially in subcortical region in asymptomatic individuals [32, 63, 64, 65, 66]. Therefore, there are wealth of previous evidence to support our findings that prevalence and proportion (i.e., number and volumes) of WMHs and ePVS associated with aging and increased cardiocerebrovascular disease risk factors (as in QRIKS3 score), and WMHs and ePVS can serve as potential biomarker of aging and cardiocerebrovascular disease risk, even among younger adults as in this study. Hence, these may assist the clinicians and health practitioners in the prevention and therapeutic strategies to reduce the burden of disability and mortality from CSVD manifestations (i.e., WMHs and ePVS) in late life.
We also found that subjects with WMHs and ePVS had lower neurocognitive performance (from WAIS-IV indices) compared with individuals without WMHs and ePVS. Alarmingly, subjects with combined lesion had much lower processing speed performance. Despite no significant association between the presence and proportion of WMHs and ePVS with any of the WAIS-IV neurocognitive indices, we did find that, the presence of ePVS was associated with reduced subjects processing speed (i.e., lower WAIS-IV PSI score). Besides, reduced hippocampal volume in ePVS subjects is also associated with reduced working memory performance based on the WAIS-IV WMI scores. Additionally, we found that aging and QRISK3 was associated with reduced WAIS-IV PSI scores.
Previous studies had suggested that individuals with CSVD manifestation such as WMHs and ePVS (with higher cardiocerebrovascular disease risk) are more vulnerable in developing neurocognitive deficits (processing speed, memory, and perceptual reasoning) [67, 68]. Moreover, Liu and colleagues suggested that, reduced white matter microstructural integrity due to WMHs at tract specifics and/or distal tracts were associated with reduced executive function and attention [69]. Previous cross-sectional, case control, and cohort studies using adjusted odd ratio reported that ePVS may adversely influenced the neurocognitive function [63, 70, 71, 72, 73, 74], particularly the speed of processing [75], which are in line with our findings with reduced processing speed and working memory (i.e., ePVS-related reduced hippocampal volume). However, some studies suggested that different ePVS locations have different manifestations, and the etiology of ePVS in different locations may differ [63, 76], which may also explain our results variability. More data are needed to further analyze the association between the ePVS regions and cognitive function (particularly the processing speed). Hence, further research delineating the role of the white matter tract involvement in WMHs and ePVS may be beneficial to better associate the presence and proportion of WMHs and ePVS with neurocognitive function outcome.
Furthermore, the variability of the results between neurocognitive profiles
(seen in WAIS-IV indices) used in our study may be due to limited sample size and
with no longer-term follow-up data. We recruited the asymptomatic individuals who
were as young as 25 years old. Chen and Li in their study (N = 142) had reported
that, about 67% of subjects (age: 20–59 years) with WMHs but not associated
with reduced cognitive function. However, 58% of the older subjects (age:
However, it is important to highlight the importance of aging and cardiocerebrovascular disease risk factors (independent of WMHs and ePVS) that may lead to degree of neurocognitive deficits [80]. Previous study had reported that 37% of individuals with higher cardiocerebrovascular disease risk factors had neurocognitive deficits [81], especially older individuals (age: 65–74 years) with hypertension [82]. Besides, long-term elevated SBP, especially in middle-to-old age individuals, has been reported as major risk factor for heightened neurocognitive deficits and dementia [48, 80]. Therefore, a prevention strategy focusing on management of cardiocerebrovascular disease risk factor especially, hypertension and SBP may be beneficial to reduce the risk of neurocognitive decline in asymptomatic individual, with or without WMHs and ePVS.
In this study, we established the interrelation of WMHs, ePVS, neurocognitive profile, aging and well-characterized cardiocerebrovascular disease risk factor (as per QRISK3 score) among a relatively young (25–62 years) sample of medically and neurologically asymptomatic adults. To the best of our knowledge, this study is the first to evaluate these multimodal interrelations in suburban southeast Peninsular Malaysia, although further follow-up and ideally multi-site study is warranted. The current study was conducted at a single center clinical setting with limited sample size (N = 54) to explore the presence and proportion of WMHs and ePVS, hence limiting the generalizability of the study results. Moreover, as part of the study inclusion/exclusion criteria, many older adults were excluded due to their high QRISK3 scores (more than 20%) and were regarded as symptomatic individuals. Finally, WAIS-IV is labor-intensive and client-demanding to complete, which better suits the research setting and is impractical for routine daily clinical use for neurocognitive screening. Nevertheless, we consolidated the relevant WAIS-IV indices for the purposes of this research to ensure comprehensiveness of the assessment.
Albeit from a single center in the suburban east coast peninsular Malaysia, the present study is the first from the region to evaluate the presence and proportion of WMHs and ePVS in asymptomatic, economically active working-aged individuals using neuroimaging and neurocognitive profiling, stratified as low-to-moderate QRISK3 scores. The study showed interrelationship between occult CVSD manifestations (ie., WMHs and ePVS) with age, QRISK3 and neurocognitive function. Although WMHs did not influence the subjects’ neurocognitive performance overall, presence of ePVS did influence subjects’ processing speed and working memory (in particular, with hippocampal ePVS and reduced hippocampal volume).
MMG, CMNCMN and MM designed the research study. MMG and CMNCMN performed the research. MM and NSI provided help and advice on the study. MMG, CMNCMN, GC, and BPKN analyzed the data. MMG and CMNCMN wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Human Research Ethics Committee at Universiti Sains Malaysia (Protocol code: USM/JEPeM/15030096). Informed consent was obtained from all subjects involved in the study.
We thank the management of the Hospital Universiti Sains Malaysia for granting permission to the investigators to use patient medical records as well as space and assets belonging to the hospital during conduction of the research. The computational work for this article was partially performed on resources of the National Supercomputing Centre, Singapore (https://www.nscc.sg).
This research was funded by Fundamental Research Grant Scheme (FRGS), Ministry of Higher Education (Grant number 203/PPSP/61771193).
The authors declare no conflict of interest.
