- Academic Editor
†These authors contributed equally.
Sodium-dependent glucose transporter-2 inhibitors (SGLT-2i) have potential hypotensive effects, enhancing the hypotensive effect of renal denervation (RDN). Spontaneously hypertensive rats (SHRs) were used to verify this hypothesis and explore the associated underlying pathways.
Seven Wistar-Kyoto (WKY) rats and 35 SHRs were divided into 6 groups. The blank WKY control (W) group consisted of all 7 WKY rats, whereas the SHRs were divided into the following groups, each containing 7 rats: sham operation (Sham), renal denervation (RDN), SGLT-2 inhibitor treatment (SGLT-2i), and the combination of renal denervation with SGLT-2 inhibitor treatment (RDN+SGLT-2i). The rats in the RDN+SGLT-2i and SGLT-2i groups were gavaged with dapagliflozin (DAPA) before RDN. The sham group was subjected to a sham operation. One-week post-operation, rat tail manometry, and echocardiography were subsequently performed, and peripheral blood inflammatory cells were detected via flow cytometry before sample collection. Following sample collection, the serum, including interleukin-6, angiotensin-II, renin, and norepinephrine, was tested via enzyme-linked immunosorbent assay. Pathological testing included Masson staining of the myocardial tissue, tyrosine hydroxylase (TH) immunohistochemistry, and Fos protooncogene (C-Fos) immunofluorescence staining of the hypothalamus tissue.
Compared with RDN alone, RDN following intragastric DAPA administration reduced systolic blood pressure (SBP) in SHRs, independent of its hypoglycemic effect. Compared with the RDN group, the pathological results of the RDN+SGLT-2i group revealed a greater improvement in the intensity of TH staining in the hypothalamus tissue, closer to the normal level of the cross-sectional area in myocardial cells. Furthermore, we observed enhanced sympathetic inhibition in the brain and a reduction in the fibrotic area within myocardial cells. Additionally, the proportions of inflammatory mononuclear cell subsets and the levels of inflammatory factors improved. Although DAPA reduced inflammation and sympathetic nerve overexcitation alone, it could not completely reverse blood pressure (BP) or cardiac function. Similarly, the alleviation of inflammation and BP reduction in RDN-treated rats were inferior to those in rats treated with RDN combined with DAPA.
Compared with RDN alone, DAPA addition before RDN can considerably reduce the BP of SHRs. The enhancement of the hypotensive effect may be attributed to the inhibition of sympathetic activity and the reduction in inflammatory reactions.
Owing to the increasing number of patients with hypertension, doctors have utilized various strategies to control blood pressure (BP) [1]. The condition of patients who have used three or more antihypertensive drugs (including diuretic ingredients) without any improvement in BP is called resistant hypertension (RH) [2]. Patients with RH cannot attain a good BP under standard medication treatment conditions.
Therefore, renal denervation (RDN) [3] has been considered for use when drugs are ineffective. Regardless of the negative results of the HTN-3 study [4], HTN-3 long-term follow-up revealed a reversal in 2022, confirming its effectiveness [5]. However, studies identifying nonconventional antihypertensive drugs to improve the hypotensive effect of RDN are lacking. Studies on sodium-dependent glucose transporter-2 inhibitors (SGLT-2i) revealed that dapagliflozin (DAPA) might have positive effects on regulating sympathetic function [6], reducing BP [7], and regulating inflammatory factors [6], which has been proven in mice, and may improve the efficacy of RDN in the therapeutic range in humans. We hypothesize that SGLT-2i enhances the therapeutic outcomes of RDN through sympathetic neuromodulation; therefore, we investigated the effect of SGLT-2i on the hypotensive effect of RDN in spontaneously hypertensive rats (SHRs) and Wistar-Kyoto (WKY) rats.
All experimental animals (7-week-old; 7 WKY rats and 35 SHRs) were purchased from Charles River Co., Ltd. in Beijing (China). The daily illumination time was 12 h. Conventional feed (Charles River Co., Ltd., Beijing, China) and distilled water were provided to the rats. SHRs were randomly divided on a random number table into SHR (S), sham operation (Sham), RDN operation (RDN), RDN combined with SGLT-2i (RDN+SGLT-2i), and SGLT-2i control (SGLT-2i) groups, with 7 rats in each group. Seven WKY rats were used as blank controls (W group). The animal study was reviewed and approved by Animal Ethics Committee of Nankai University. The ethics approval number is 2022-SYDWLL-000273. The study was conducted in accordance with the local legislation and institutional requirements.
After acclimatization, the BP, body weight, echocardiogram, and flow cytometry-based monocyte population of the rats were measured at baseline. The rats were then fed until they were 12 weeks old, and their BPs were measured to ensure a hypertensive state. DAPA (1 mg/kg, dissolved in distilled water, once a day, AstraZeneca Pharmaceuticals LP, Mount Vemon, IN, USA) was subsequently gavaged into the RDN+SGLT-2i and SGLT-2i groups of rats for 1 week until the RDN operation was performed in the RDN and RDN+SGLT-2i groups (the renal artery and vein were separated after laparotomy; phenol was used for RDN treatment). A sham operation was performed in the sham group (only the renal artery was separated after laparotomy). One week after surgery, no special treatment was given to any group to alleviate stress and they were prepared for the final pressure measurement. Next, all related indicators were measured at 1 week post-operation. Samples were collected, and the serum, including interleukin-6 (IL-6), angiotensin-II (Ang-II), renin, and norepinephrine (NE), was subjected to enzyme-linked immunosorbent assay (ELISA). Pathological testing included tyrosine hydroxylase (TH) immunohistochemical staining of the renal artery and Fos protooncogene (C-Fos) immunofluorescence staining of the hypothalamus. Masson staining was performed on the myocardial tissue.
After warming the fixator with a thermal pad (CODA® Monitor, Kent Scientific, Torrington, CT, USA) for 30 minutes (CODA® Monitor, Kent Scientific, Torrington, CT, USA, the temperature was stabilized at 37 °C), we slowly placed the rats into the fixator and measured their blood pressure via a tail manometer (CODA® Monitor, Kent Scientific, Torrington, CT, USA). Each rat was subjected to 15 cycles, and then, we took the average of 3 valid values as the blood pressure for this measurement (status shown OK in Kent software, Torrington, CT, USA). The quality control of the pressure measurement images was performed by Kent’s engineers.
A depilatory cream was used to remove the fur from the chest of the rats in a quiet state, and 1.5% isoflurane (Hangzhou Yipin Pharmaceutical, Hebei, China) was used for maintenance anesthesia. An animal echocardiogram (VisualSonics Vevo 2100, Toronto, Canada) was used in the cardiac module. After the location of the heart was determined, the cardiac ejection fraction (EF) and other data were measured via the short axis section of the sternum.
First, approximately 50 µL of blood was taken from the tail vein of each
rat and added to an ethylenediaminetetraacetic acid (EDTA, Sigma-Aldrich, MO,
USA) anticoagulant blood collection tube. Second, 0.25 µL of PerCP/cyanine
5.5-conjugated anti-CD43 antibody (100 uL, 202818, BioLegend, San Diego, CA, USA),
0.25 µL of PE-conjugated anti-rat-CD172a antibody (100 uL, 204706,
BioLegend, San Diego, CA, USA) and 50 µL of anticoagulant blood were mixed
evenly in the tube and incubated at room temperature for 15 minutes in the dark.
After that, 1 mL of red blood cell lysis buffer (C3702, Beyotime, Beijing, China)
was added to the tube, which was subsequently incubated at room temperature in
the dark for 10 minutes and then centrifuged at 350
We used FL2 and FL4 of a BD Accuric C6 Plus flow cytometer (BD, Franklin Lakes Campus, NJ, USA) to collect fluorescence signals (log logarithmic mode). Owing to the high expression of CD172a in rat monocytes, monocytes can be circled according to the different fluorescence intensities of CD172a. With SSC-Log as the ordinate and CD43-PE-Cy5-Log as the abscissa, peripheral blood mononuclear cells were divided into two subgroups: classic CD172a+CD43+ (CD43 low) and non-classic CD172a+CD43++ (CD43 high) (according to the different fluorescence intensities of CD43 expressed by cells). BD Accuric C6 Plus software (The system comes with built-in software, BD, Franklin Lakes Campus, NJ, USA) was used to analyze the proportions of the different cell subgroups after the gate was set.
ELISA was carried out according to the instructions of the kits. (All kits Purchased from Elabscience, Wuhan, China).
We used depilatory cream to remove fur from the abdomens of the rats, which were in a quiet state, and then induced with general anesthesia via isoflurane (3% isoflurane, purchased from Hebei Yipin Pharmaceutical, Hebei, China; 97% oxygen) at a concentration of 1.5% for maintenance anesthesia. The warming pad held the temperature at 37 °C. The abdominal cavity was opened along the midline of the abdomen via a scalpel, and the renal artery and vein were separated after the renal artery was located along the aorta. Then, 10% phenol solution was used for ablation under an operating microscope (phenol purchased from Sigma‒Aldrich, MO, USA). During the ablation process, phenol solution was used to smear the renal arteries for 2 minutes, whereas physiological saline was used to smear the renal veins. After both renal artery procedures were completed, it was confirmed under the microscope that the ablation was successful again. Finally, the muscle and skin were sutured layer by layer. In the sham group, after the abdominal cavity was opened and the arteriovenous sheath was exposed, only physiological saline was applied.
Method of euthanasia and the anesthetics: At the end of the experiment, the mice were anesthetized with 3% isoflurane and then euthanized in a CO₂ chamber. Blood and aorta samples were then collected. Renal denervation (RDN) was performed as previously described. After induction of general anesthesia using isoflurane, the mice were placed in a supine position on a platform, and an abdominal incision was made.
Before pathological testing, the tissues were fixed with 4% paraformaldehyde. All fields subjected to pathological staining were randomly selected, and the photographer was blinded. ImageJ 6.0 (LOCI, University of Wisconsin, Madison, WI, USA) was used for analysis.
Masson’s trichrome staining was conducted via a Masson Kit (1.00485, Sigma-Aldrich, MO, USA). The tissue sections (thickness = 5–6 µm) were cut and placed on microscope slides (LeicaRM2235, Germany).
The slides were heated at 60 °C for 2 h, after which xylene was used for dewaxing. Each slide was hydrated for 3 minutes with gradient ethanol (anhydrous ethanol, 95%, 90%, 80%, and 70% ethanol; all ethanol was purchased from Shandong Bocheng Chemical Co., Ltd., Shandong, China). The slides were rinsed with running water for 5 min and then rinsed with deionized water once. The slides were immersed in Bouin’s solution (HT101128, Sigma-Aldrich, MO, USA) at 56 °C for 15 minutes. The slides were rinsed with deionized water for 10 minutes until the yellow color disappeared and then stained with Weigert’s hematoxylin (1.00485, Sigma-Aldrich, MO, USA) for 5 minutes. After being washed with deionized water, each slide was differentiated with 1% hydrochloric acid alcohol differentiation solution for 10 seconds. The samples were then washed with running water for 10 min. Next, the slides were stained with Biebrich scarlet-acid fuchsin (1.00485, Sigma-Aldrich, USA, MO, USA) for 5 minutes and then washed again with deionized water twice. Then, we incubated the slides in 2.5% phosphotungstic-phosphomolybdic acid (1.00485, Sigma-Aldrich, USA, MO, USA) for 5 minutes. All the slides were dyed with aniline blue for 5 minutes and fixed in 1% acetic acid for 1 minute. Finally, the slides were rinsed with distilled water, dehydrated with anhydrous ethanol for 2 minutes and mounted with neutral resin (10004160, Shanghai National Pharmaceutical Group Chemical Reagent Co., Ltd. (SCRC), Shanghai, China).
The hypothalamus was excised, fixed (paraformaldehyde, 4.0%), paraffin-embedded, and sectioned into 3 µm slides. The sections were deparaffinized, and antigen retrieval was performed. The slides were incubated with rabbit polyclonal anti-tyrosine hydroxylase (1:300, Millipore AB152, Sigma-Aldrich), followed by incubation with goat anti-rabbit IgG (1:300, GB21303, HRP-marked, Servicebio, Wuhan, China).
In a blinded fashion, staining intensity (degree of tyrosine hydroxylase
staining) was scored as negative, marked 0; 1, weak (blush); 2, mild; 3,
moderate; or 4, strong. The proportion of nerves showing decreased tyrosine
hydroxylase staining was scored as follows: 1,
All slides of the hypothalamus were fixed (paraformaldehyde, 4.0%), paraffin-embedded, and sectioned into 3 µm slides. The sections were deparaffinized, and antigen retrieval was performed. We used EDTA antigen repair solution for repair (medium fire for 8 minutes, no fire for 8 minutes, and transition to medium low fire for 7 minutes). We incubated the slides with PBS, 1% donkey serum, and primary mouse anti-SGLT-2 antibody (1:500, Servicebio, Wuhan, China) at 4 °C overnight. After overnight incubation, we washed the slides in PBS (pH 7.4) 3 times for 5 min each, transferred them to a solution containing PBS, 1% donkey serum, and a secondary generic antibody (goat anti-mouse IgG marked with CY3, 1:10, Servicebio, Wuhan, China) and incubated them for 1 h thereafter, after which they were rinsed once for 5 min with PBS. Next, we incubated the slides with DAPI at room temperature for 10 minutes to stain the nuclei. Finally, the slides were rinsed with distilled water, dehydrated with graded ethanol, cleared with turpentine oil and coverslipped with neutral balsam.
The slides were heated at 60 °C for 2 h, after which xylene was used for dewaxing. Gradient ethanol (anhydrous ethanol, 95%, 90%, 80%, and 70% ethanol; all ethanol was purchased from Shandong Bocheng Chemical Co., Ltd., Shandong, China) was hydrated for 3 minutes each. The samples were rinsed with running water for 10 minutes and then soaked in deionized water for 10 minutes. At room temperature, the slides were washed twice with WGA incubation solution (WGA purchased from Sigma-Aldrich, L4895, MO, USA, 1:100 diluted with 1 mmol of hydrochloric acid) on an oscillator for 5 minutes each. After the samples were soaked in PBSC for 30 minutes, we used absorbent paper to absorb the excess liquid. Then, we added WGA working solution (1:100 diluted with 1 mmol of hydrochloric acid) to the slides (30–50 µL per slide). We placed the slides in a refrigerator at 4 °C and incubated them for 30 minutes in the dark and then precooled them with precooled PBS in the refrigerator for 30 minutes. Next, we washed the slides in precooled PBS on an oscillator 3 times, each time for 5 minutes. Then, the excess liquid was absorbed with absorbent paper, and 30–50 µL of DAPI working solution (DAPI, G1012, Servicebio, 1:100 dilution, the diluent was the antibody mixture; U3635, Sigma-Aldrich, MO, USA) was added dropwise to each slide. The slides were incubated at room temperature in the dark for 10 minutes, after which they were washed again with precooled PBS. Finally, the slides were covered with 50% buffer glycerin.
All the data are expressed as the mean
There was no difference in systolic blood pressure (SBP) (Fig. 1C), body weight,
or blood glucose at baseline. However, both RDN+SGLT-2i and SGLT-2i groups showed
a significant decrease in body weight compared with the W group (RDN+SGLT-2i:
254.00
 Fig. 1.
Fig. 1.
Body weight, blood glucose, and blood pressure (BP). (A) Body
weight after treatment. (B) Blood glucose after treatment. (C–E) Systolic blood
pressure from baseline (C), second measurement (12 weeks old) (D) to the endpoint (E). The
data are presented as the mean
The SBP in S and Sham group of rats (12-week-old) increased significantly vs WKY rats (p
SBP in the RDN+SGLT-2i group was lower than that in the RDN or SGLT-2i group under combination therapy (p
Inflammatory cells reflect real-time changes in inflammatory activity, with
monocytes showing the most notable changes during the progression of
hypertension (Fig. 2A–F). Changes in monocytes were detected by flow cytometry, and no
significant differences between monocyte subsets were found at baseline. However,
before sampling, the proportion of clusters of differentiated CD43+ cell
subsets in the S or Sham group was significantly greater than that in W group (Fig. 2G, p
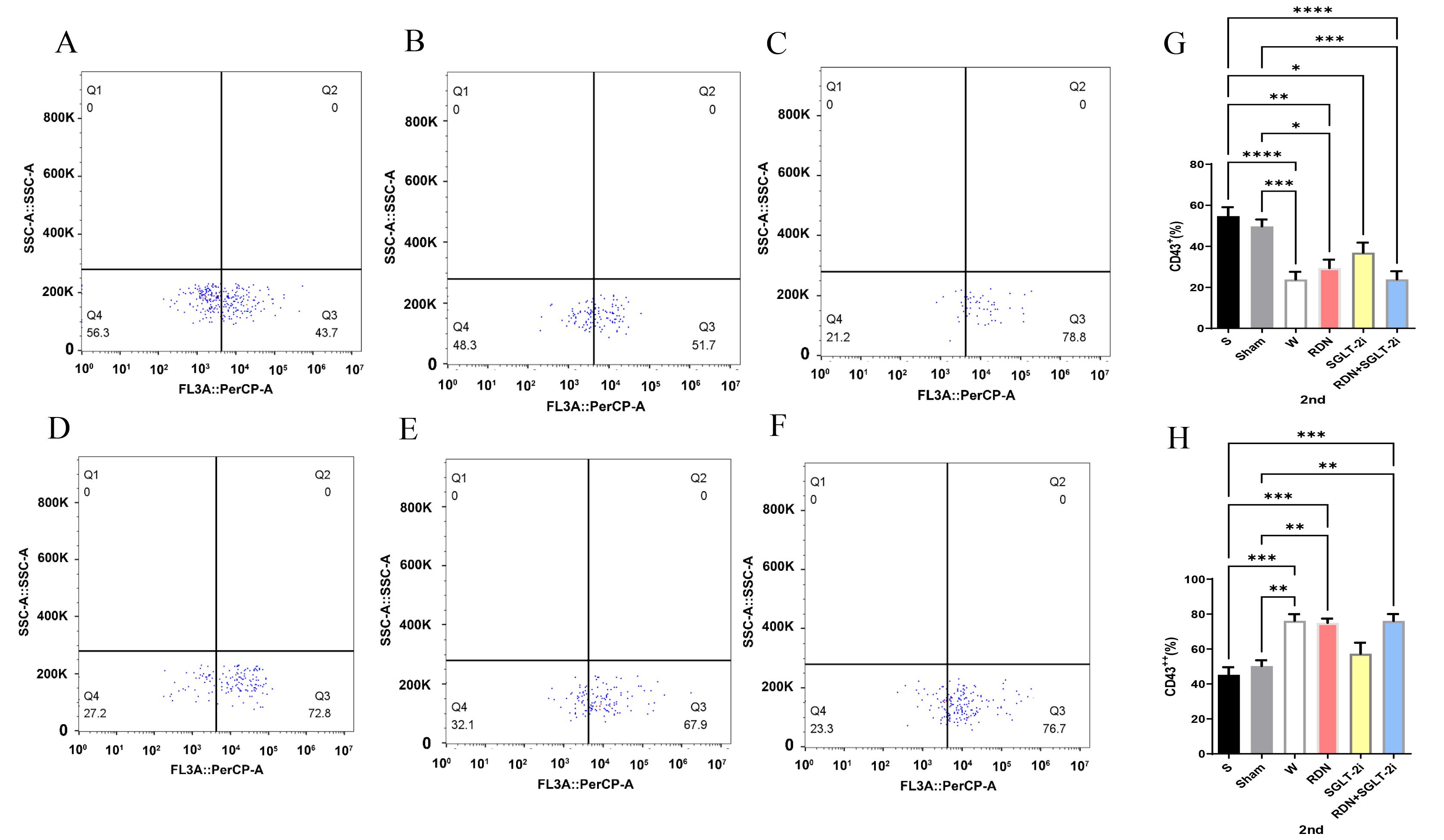 Fig. 2.
Fig. 2.
Changes in the proportion of monocytes. (A–F) Scatter plots
from flow cytometry (A) S group; (B) Sham group; (C) W group; (D) RDN
group; (E) SGLT-2i group; (F) SGLT-2i+RDN group. (G–H) Proportion of flow cytometry
subpopulations (G) proportion of CD43+ monocytes at the
endpoint; (H) proportion of CD43++ monocytes at the endpoint. The data
are presented as the mean
Similarly, the S and Sham groups presented
the lowest proportions of CD43++ monocytes, significantly lower than that in W group (Fig. 2H, p
There was no significant difference in the ejection fraction (EF) among the
groups when an echocardiogram was performed at the baseline. Before sampling, EFs
in the S and Sham groups were significantly lower than that in the W
group (p
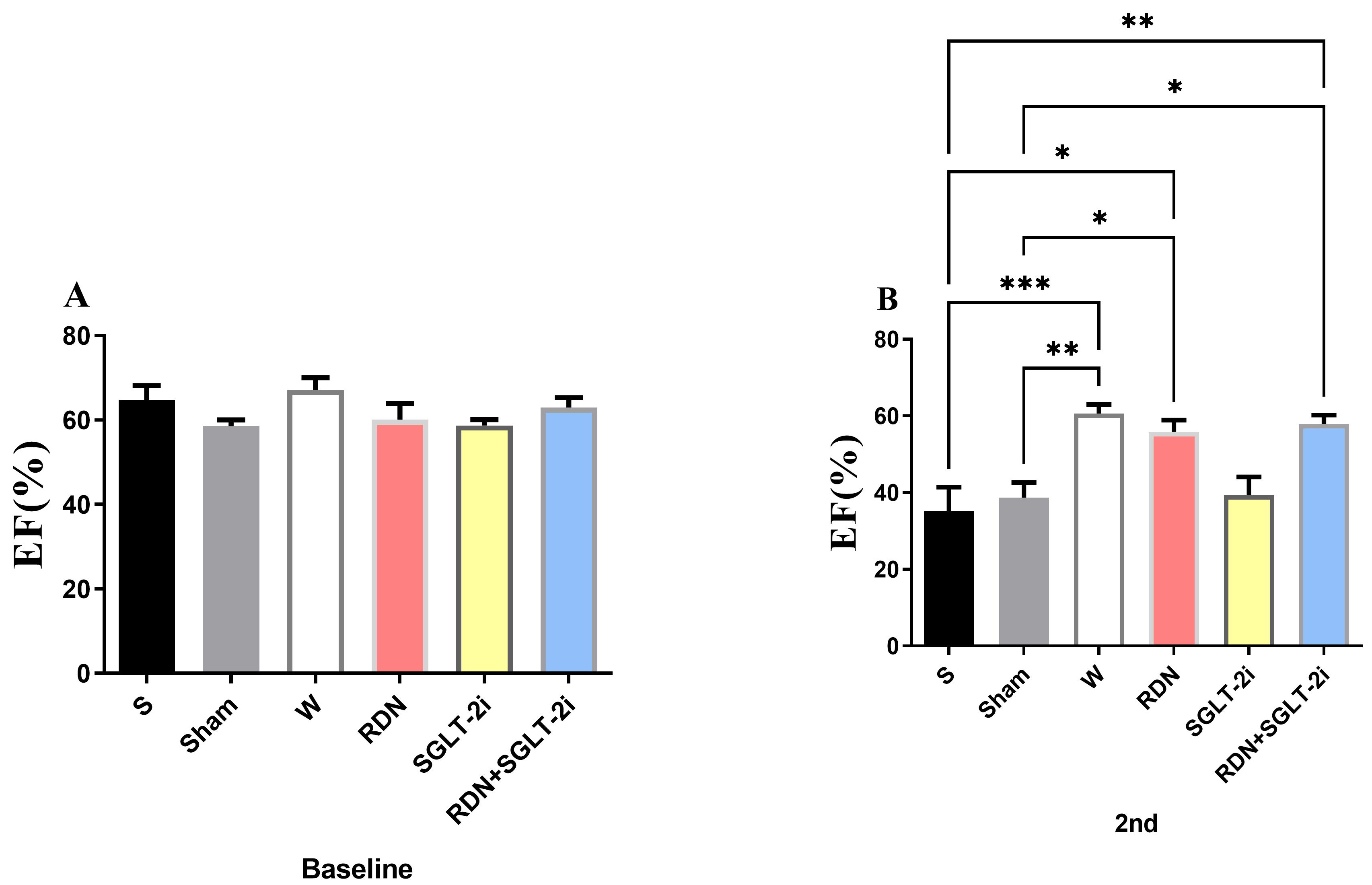 Fig. 3.
Fig. 3.
Ejection fraction alteration in the echocardiogram. (A)
Baseline EF; (B) EF at the endpoint. One-way analysis of variance, *p
The contents of IL-6, Ang-II, NE, and renin in the rat serum were detected
through ELISA. No significant differences were found in the IL-6 levels between
the S and Sham groups (Fig. 4B), but the IL-6 levels in S groups were
significantly greater than those in the other groups (p
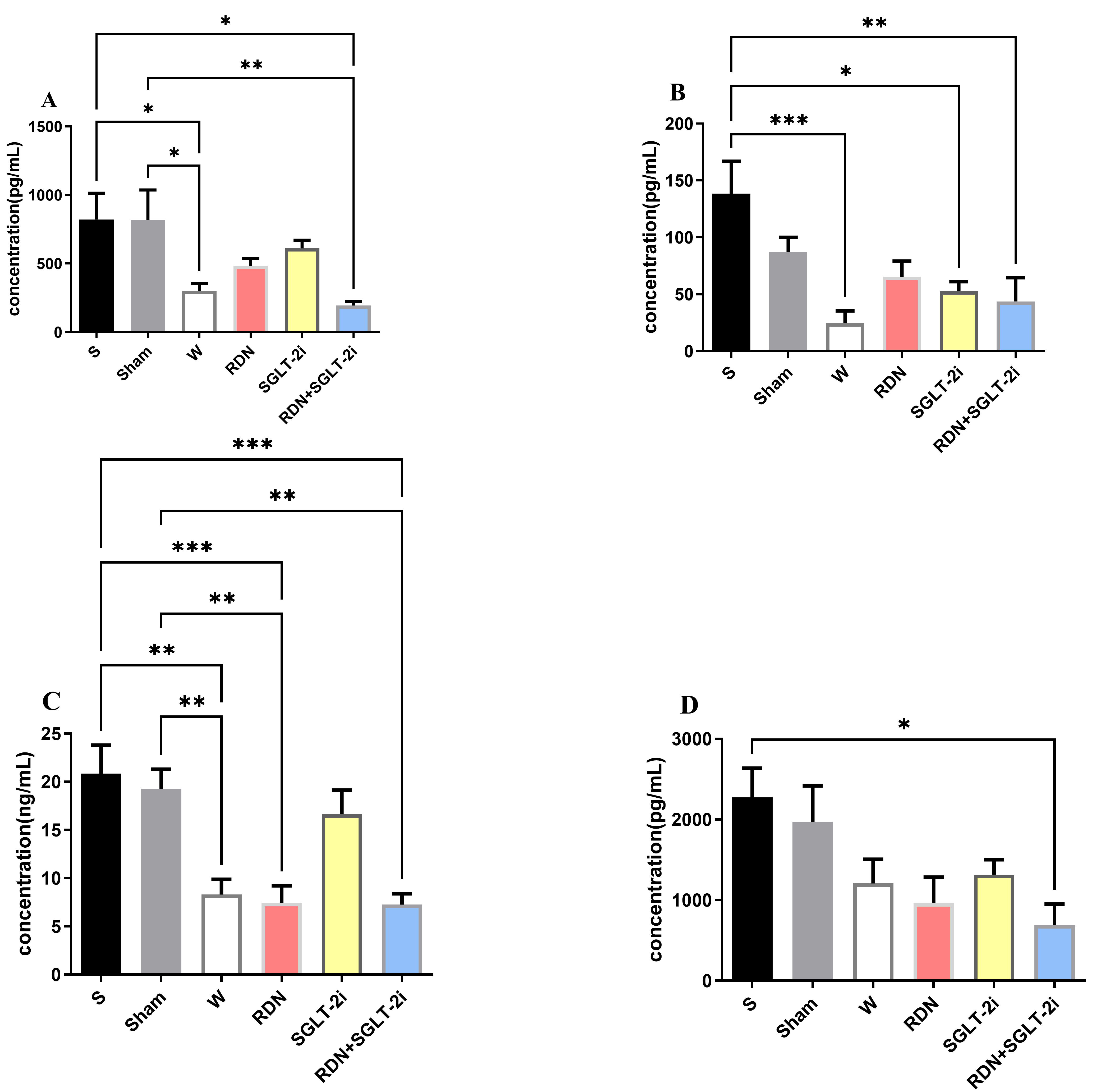 Fig. 4.
Fig. 4.
Enzyme-linked immunosorbent assay analysis of inflammatory
factors in rats. The concentrations of (A) Renin; (B) IL-6; (C) NE; and (D)
Ang-II in the rat serum. One-way analysis of variance. The data
are presented as the mean
Masson staining was performed to further understand the degree of myocardial
fibrosis (Fig. 5A–F). Notably, the myocardial fibrosis area in the S group was
not significantly different from that in the Sham group or the SGLT-2i group. The
percentage in the RDN+SGLT-2i groups was significantly lower than that in
the S/Sham group (p
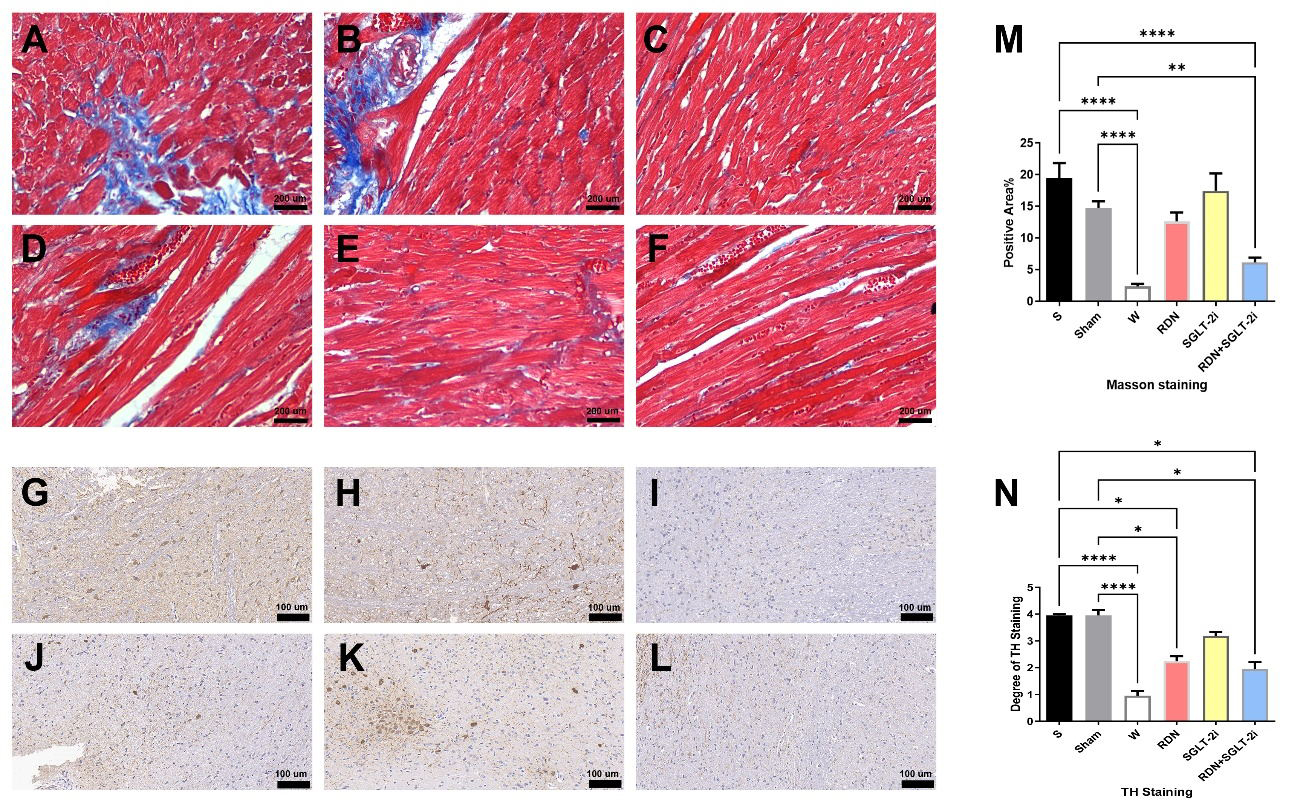 Fig. 5.
Fig. 5.
Masson and TH staining (200
C-Fos, an early immediate memory gene, reflects the activity of the
sympathetic nervous system, and its high expression represents the inhibition of
sympathetic hyperexcitability (Fig. 6A–F). The visible average optical density
(AOD) in the RDN+SGLT-2i group was greater than that in the other groups (p
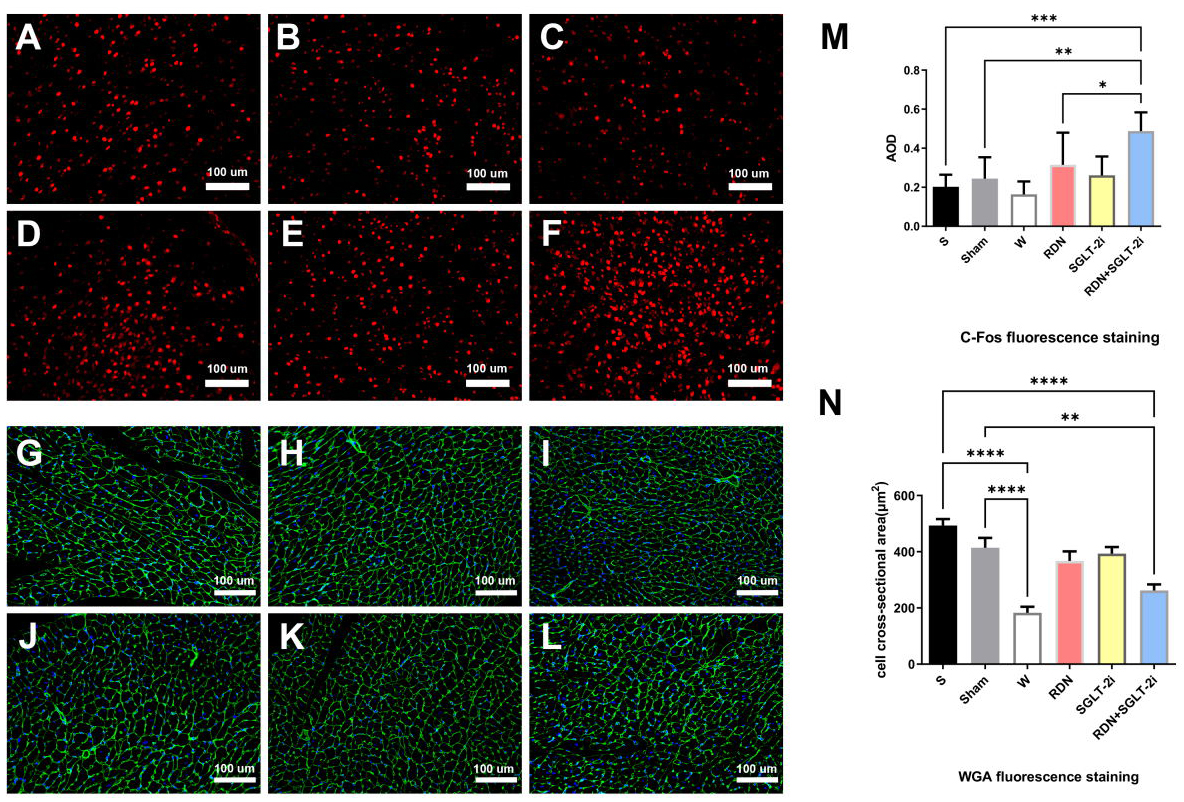 Fig. 6.
Fig. 6.
C-Fos and WGA fluorescence staining. (A–F)
C-Fos fluorescence staining (200
Wheat germ agglutinin fluorescence staining revealed a cross-sectional area of
cardiomyocytes (Fig. 6G–L). The results revealed considerable cardiomyocyte
hypertrophy in both the S and Sham groups, and there was no significant
difference in the cross-sectional area of cardiomyocytes between these two groups.
Compared with that in the W group, the cross-sectional area in the S and Sham
groups was significantly greater (p
Notably, adding DAPA before the RDN operation clearly enhanced
the antihypertensive effect. However, this enhancement was not correlated with BP
or blood glucose at the final measurement (Pearson correlation
coefficient: r
The antihypertensive effect of DAPA in enhancing RDN operates independently of its hypoglycemic mechanism. Further evidence suggests that DAPA can regulate central sympathetic nerve activity independently of its hypoglycemic effects. Reportedly, DAPA treatment alters C-Fos expression in the hypothalamus of mice, which is correlated with central sympathetic nervous system activity [6]. More studies have directly shown that SGLT-2i alters the activity of the sympathetic nervous system, not only in animal experiments [9], but also in clinical studies with similar results [10, 11]. Our results are consistent with these findings, suggesting that C-Fos expression in the hypothalamus is indicative of its regulatory effect on sympathetic nerves, an effect modulated by DAPA.
We measured the EF values of the rats and found that the cardiac function in the RDN+SGLT-2i group was better than RDN group. These findings suggest that our hypothesis may be correct, which is further consistent with the pathological results. Immunohistochemical staining revealed that the scores of the RDN+SGLT-2i and RDN groups were markedly lower than S group. Notably, the EF tended to decrease in the S and Sham groups, which is also consistent with the pathological results. RDN+SGLT-2i group had a better EF than the RDN group, indicating that DAPA combined with RDN is useful for improving hypertension-induced cardiac insufficiency. The results of TH staining suggested that the higher the C-Fos expression in the hypothalamus was, the lower the degree of fibrosis in the myocardial tissue was, which confirmed the success of RDN. Therefore, DAPA affects BP through the central sympathetic nervous system. However, this did not completely explain all the issues.
A decrease in the weight of the rats was observed after DAPA treatment, which was more pronounced in the RDN+SGLT-2i group. Weight loss is considered one of the contributing factors to antihypertension [12, 13]. Reportedly, the DAPA group lost more weight in randomized controlled studies of the population than did the control group [14]. However, the hypotensive effect seems not to be simply explained by weight loss.
C-Fos expression in the RDN group was considerably increased, indicating a notable improvement in overexcitation of the sympathetic nervous system after RDN treatment. Furthermore, the high C-Fos expression in the RDN+SGLT-2i group confirmed our initial hypothesis that combining these two therapies was feasible. Combining the cardiac function indicators, we found that the pathological results were consistent with those of the cardiac function indicators in the RDN+SGLT-2i group. Pathology revealed a smaller cross-sectional area of myocardial cells via WGA fluorescence staining, increased C-Fos fluorescence intensity, and a lower percentage of myocardial fibrosis in the RDN+SGLT-2i group than in the control group, indicating that cardiac function was protected by the combined treatment of RDN and DAPA. The EF of the RDN+SGLT-2i group rats was similar to the W group. Additionally, the difference between the RDN+SGLT-2i and S groups was more significant based on the comparison of TH staining, which was better than that of the RDN group. This decrease in inflammatory factor levels is consistent with the pathology and ultrasound results.
Inflammatory reactions are highly important in the process of hypertension
[15, 16, 17]. The ELISA results confirmed that the IL-6 levels in the RDN+SGLT-2i or SGLT-2i group was
markedly lower than those in the S group,
suggesting the involvement of the upstream Toll-like receptor (TLR) pathway.
Evidence of increased IL has also been reported in studies on salt-sensitive
hypertension [18]. Renin and Ang-II levels exhibited similar trends. Studies have
reported the anti-inflammatory effect of RDN [19, 20, 21]. Considering that DAPA can
exhibit an inhibitory effect on inflammatory factors alone, combination treatment
with DAPA and RDN might involve a common pathway for inhibiting inflammation. In
recent study, DAPA has been shown to reduce inflammatory reactions via the TLR
pathway, possibly by affecting the expression of microRNAs, which promote
inflammation in cells and macrophage transformation [22]. The association of
monocyte clustering with the inflammatory state in the body and heterogeneity in
monocyte subsets were discovered more than a decade ago [23]. The monocytes of
rats can be divided into two subgroups, namely, CD43++ and CD43+
subsets, which reflect their proinflammatory and anti-inflammatory biological
effects, respectively. An increase in the number of proinflammatory CD43+
cells was observed, which was most notable in the S and Sham groups. There was a
significant difference between the SGLT-2i and S groups, and the difference
between the RDN+SGLT-2i and S groups was more significant, indicating that the
combination treatment of RDN and DAPA considerably reduced the proportion of the
proinflammatory subset of monocytes. ELISA and flow cytometry results indicated
the close relationship between the nuclear factor kappa B
(NF-
Theoretically, DAPA has a definite therapeutic effect on heart failure [26, 27], but our results contradict present theories. The pathological results, especially the Masson staining results, revealed no remarkable improvement in the proportion of patients with myocardial fibrosis in the SGLT-2i group. Moreover, the AOD from C-Fos fluorescence staining suggested that SGLT-2i may regulate sympathetic nerve activity, but its AOD did not increase considerably compared with that of the S group. This result indicated that the ability of the SGLT-2i group to regulate the sympathetic nervous system was notably lower than that of the RDN+SGLT-2i and RDN groups. The pathological staining results were consistent with those of EF. These findings suggest that short-term DAPA use may affect sympathetic nervous activity to some extent but that it cannot completely improve myocardial injury or cardiac function. This speculation did not contradict the present guidelines. Interestingly, the AOD of the S and Sham groups were slightly greater than that of the W group according to C-Fos fluorescence staining. There may be a neurofeedback mechanism targeting sympathetic overactivation, similar to the higher AOD values in the RDN+SGLT-2i group, but we have not been able to further explore this. Suggesting that SHRs might compensate for hypertension via their sympathetic neuromodulation mechanism and that they could not fully resist hypertension-induced harm.
Overall, DAPA combined with RDN enhanced the antihypertensive effect of RDN and improved the anti-inflammatory response and cardiac function. This was evidenced by the inhibition of the central sympathetic nervous system, and inflammatory reactions. The reported positive effects on BP and cardiac dysfunction may have clinical significance for hypertension patients with heart failure. Further studies are needed to explore the underlying mechanisms and conduct clinical research in patients with hypertension.
The original contributions further inquiries can be directed to the corresponding authors.
HZ & ZL: Writing—original draft, conceptualization, methodology; CL, YL: writing—review & editing, conceptualization, methodology, project administration; WC: writing—review & editing, conceptualization, methodology; YW: writing—original draft, conceptualization, methodology; JF: writing—review & editing, formal analysis; XY: methodology, writing—review & editing; QL: methodology, writing—review & editing; NY: methodology, writing—conceptualization, review & editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The animal study was reviewed and approved by Animal Ethics Committee of Nankai University. The ethics approval number is 2022-SYDWLL-000273.The study was conducted in accordance with the local legislation and institutional requirements.
Not applicable.
This work was supported by grants from the National Natural Science Foundation of China (NO. 82300445 and 82470294), Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-054B), and Natural Science Foundation of Tianjin (22JCQNJC01130). We are grateful for this generous funding.
The authors declare no conflict of interest.
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
