1 Department of Anesthesiology, Renmin Hospital of Wuhan University, 430060 Wuhan, Hubei, China
†These authors contributed equally.
Academic Editor: Kebin Hu
Abstract
Background: Circadian rhythms are fundamental to regulating metabolic
processes and cardiovascular functions. Phosphorylated PERIOD2 (PER2) is a key
factor in determining the period of the mammalian circadian clock. Moreover,
casein kinase 1
Keywords
- PER2
- CK1ε
- phosphorylated PER2
- PF-670462
- diabetic myocardial injury
Diabetes mellitus (DM) is a lifelong metabolic disease and its prevalence continues to increase [1, 2]. DM affects myocardial morphology, metabolism, gene expression, physiological function, and much more. Diabetes-associated myocardial structural and functional changes are known as diabetic cardiomyopathy [3]. Cardiovascular complications caused by diabetic myocardial injuries are among the major causes of mortality and morbidity in diabetic patients. However, diabetes’ exact pathogenesis is complicated, and there is no effective treatment method for the condition yet. Therefore, actively exploring its potential mechanism could provide a useful protective measure to reduce the incidence of diabetes-related complications.
In mammals, rhythms are orchestrated by a master time-keeping mechanism housed within the suprachiasmatic nuclei (SCN) of the hypothalamus, and peripheral organs, such as the heart, have their individual self-endogenous clocks as well. Myocardial contraction, relaxation, and effective blood pumping exhibit rhythmic fluctuations [4, 5, 6]. Increasing evidence suggests that diabetes inter-relates with disrupted circadian clock rhythms [7, 8, 9, 10]. The circadian clock consists of multiple core proteins, including BMAL1, ClOCk, PERIOD (PER1-3) and CRYTOCHROME (CRY1-2), that interact to influence each other’s transcription and function. Of these proteins, PER2 is enormous, with a well-defined N-terminal region containing PER-ARNT-SIM (PAS) domains and two unique competing phosphorylation sites [11, 12, 13]. These motifs and domains are responsible for multiple homodimeric and heterodimeric protein interactions, as well as PER’s stability, cellular localization, and inhibitory activity toward CLOCK:BMAL [14, 15, 16]. Most of all, PER2 plays a significant role in the integration of signals and helps to robustly compensate for profound changes that would disrupt the circadian clock. Additionally, PER2 in the heart has a notable impact on its gene expression and overall tissue function [7, 17, 18]. Studies have shown that diabetes causes PER2 to exhibit massive phase shifts in mRNA levels and severely aggravated myocardial functional disorders [19, 20]. It has also been demonstrated that pharmacologically targeting PER2 improves glucose homeostasis in obesity [8]. These findings collectively indicate that myocardial pathological processes during diabetes are closely associated with the clock gene PER2. However, changes in the PERIOD2 (PER2) protein forms and their exact mechanisms in diabetic myocardial injuries remain unclear.
Casein kinase 1 (CK1), a member of the superfamily of serine/threonine-specific
kinases, participates in modulating various cellular activities, such as
circadian rhythm and glycolipid metabolism [21, 22, 23]. CK1
To explore the adaptive connection between diabetes and PER2, we have
highlighted the exact impact of diabetes on PER2 and CK1
Male Sprague-Dawley rats (8-weeks old, weighing
180~220 g) were obtained from Beijing Vital River Laboratory
Animal Technology Co, Ltd and maintained in an SPF environment at an ambient
temperature of 20–22
PF-670462 was purchased from Topscience,
and its potency (IC50 = 7.7
After adaptive feeding for several days, type-1 diabetes was generated in rats by intraperitoneally injecting animals with streptozotocin (STZ, Sigma, 65 mg/kg) dissolved in citrate buffer, as described previously [32, 33, 34]. Rats in the non-diabetic group were injected with the same amount of citrate buffer. 72 hours after STZ administration, blood was drawn from the tail vein of each rat once to measure fasting blood glucose (FBG). Rats exhibiting hyperglycemia (blood glucose levels higher than 16.7 mmol/L) were considered to have diabetes and continuously fed for at least 8 weeks. FBG and body weight were monitored weekly.
All animals were synchronized to a fixed 12 h light/12 h dark schedule (L/D cycle) — ZT0 (Zeitgeber Time) was set at 7:00 AM, and ZT12 at 7:00 PM — and placed in their home cages, with lighting set at 250 to 300 lux for each box. Their individual running periods and spells of activity onset were free of restriction and recorded daily.
At the end of the scheduled
course, rats were tranquilized, and their apical blood was collected in
anti-coagulation tubes. Heart tissues were rapidly excised and washed with cold
PBS and 0.9% Nacl. These heart tissues and blood samples were collected at ZT5,
ZT11, ZT17, and ZT23 (n = 4/time point/group). Also, randomly chosen diabtic rats
were treated with or without PF-670462 (30 mg/kg, i.p., n = 5/group) once at ZT11
and sacrificed at ZT23. Zeitgeber Time and dosage were applied as per previous
dose-response rates [21, 29, 31], and all collected samples were stored at –80
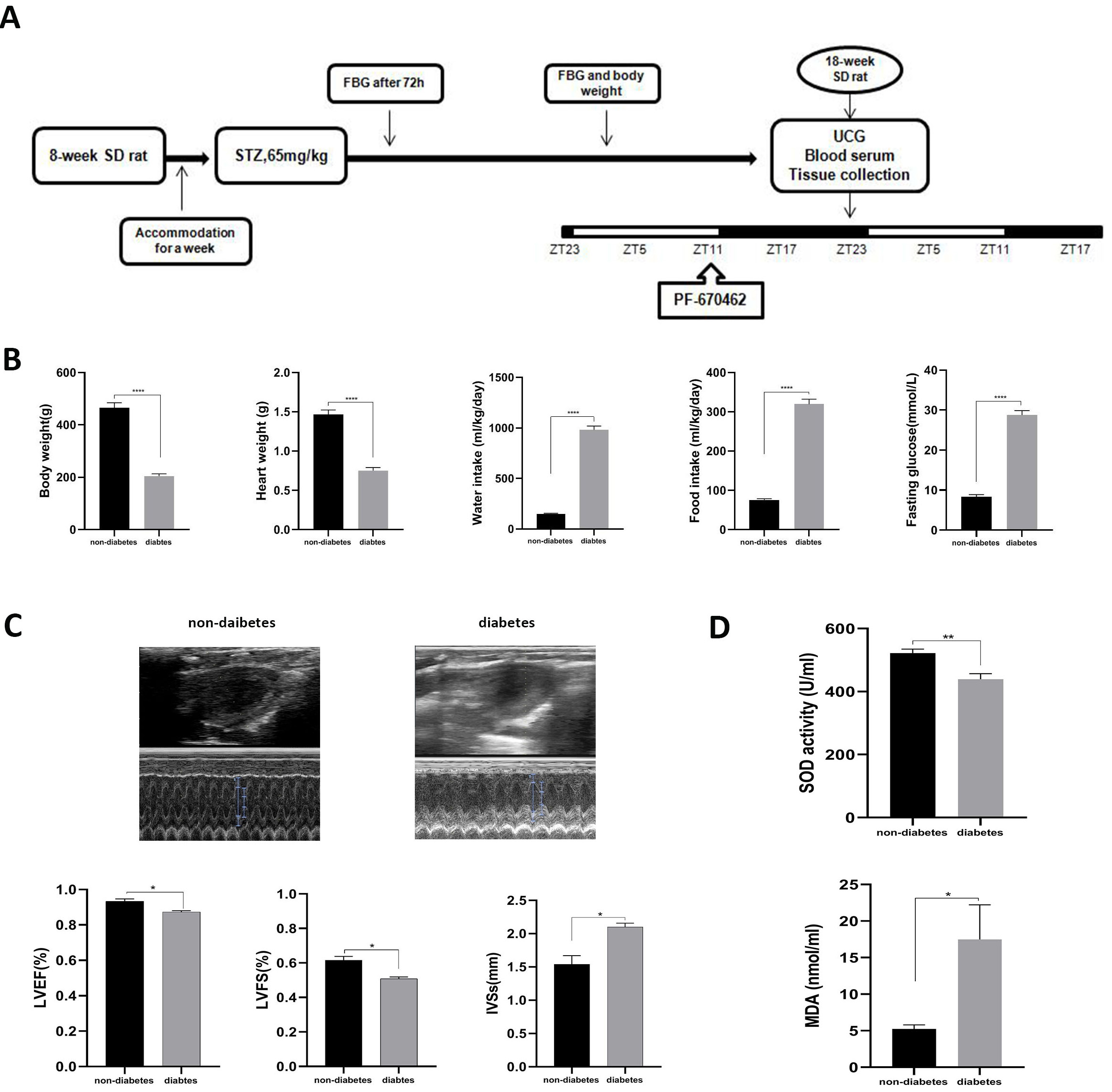
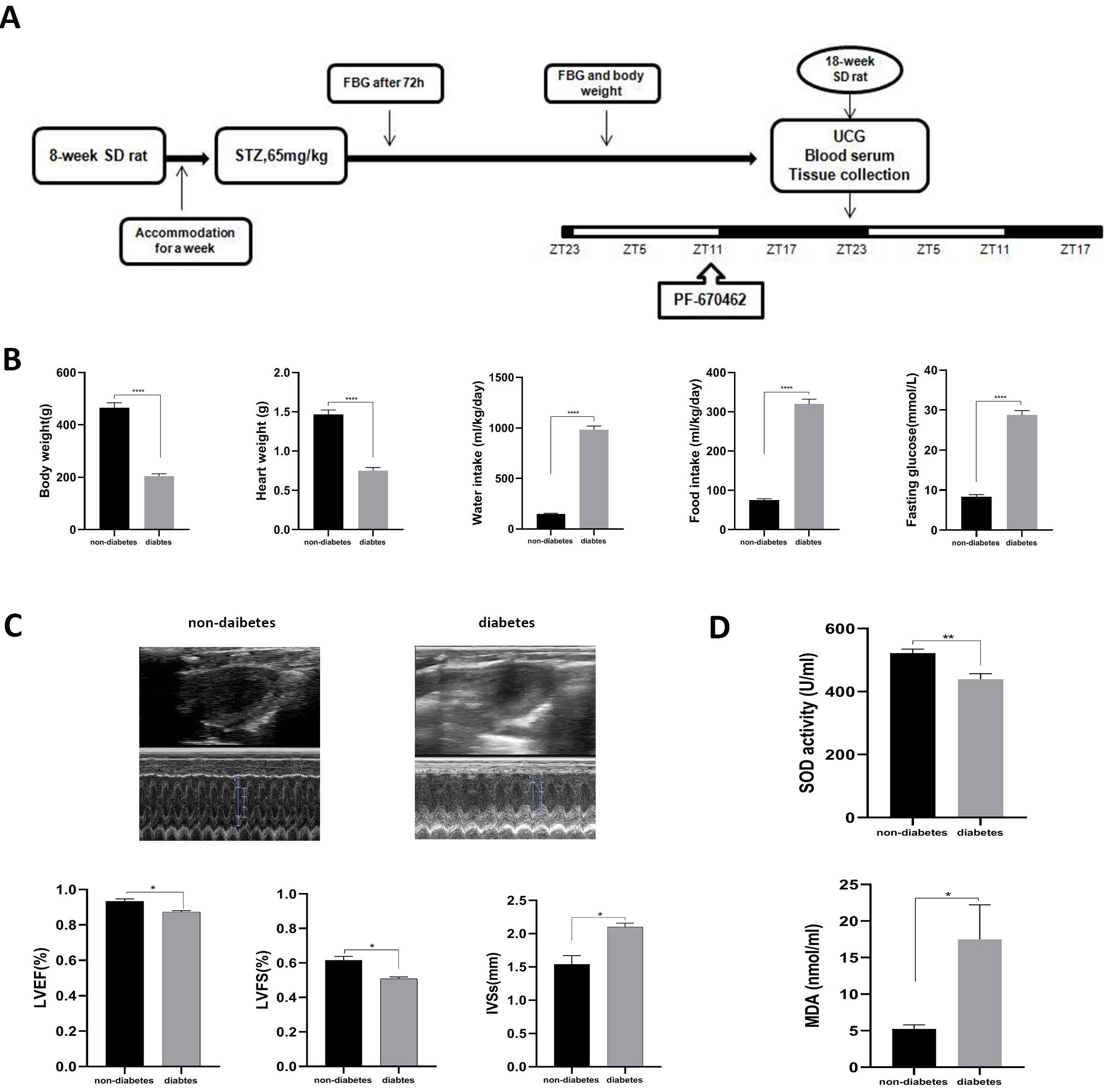 Fig. 1.
Fig. 1.General characteristics and the myocardial injury in STZ-induced
diabetic rats. (A) Showed the general experimental design. (B) Showed the
animals’ genecral characteristics. (C) Showed the cardiac function measured by
ultrasound cardiogram (UCG). (D) Showed the indicators of oxidation stress
injury. Data was expressed as the Mean
Cardiac function was monitored using an animal ultrasound cardiogram (UCG) to measure parameters, including IVSs, left ventricular EF%, and FS%. Two-dimensional and M-mode echocardiographic measurements were analyzed with a GE vivid 7 E95 high resolution in vivo-imaging system (VisualSonics, Toronto, ON, Canada).
Rat heart tissues were homogenized
in TriReagent (Sigma) using a Liquid nitrogen grinding instrument (Mo Bio
Laboratories Inc, Carlsbad, CA), and total RNA was isolated according to the
manufacturer’s instructions and reverse transcribed. SYBR Green qPCR was then
applied to the reverse transcription product in real-time quantitative RT-PCR
(Applied Biosystems 7500 Real-Time PCR System). The expression of genes within
each sample was normalized against
Blood glucose was determined using a portative glucometer, MediSense Optium Xceed (Abbott Diagnostics Ltd, Maidenhead, UK). The biochemical marker of myocardial injury, LDH, was assayed using an automatic biochemical analyzer (SIEMENS, ADVIA 2400, Germany).
Myocardial injury was quantified using a TdT-mediated dUTP nick-end labeling (TUNEL) technique in in situ slices utilizing a One Step TUNEL Apoptosis Assay Kit (Beyotime, Jiangsu, China) according to the manufacturer’s instructions. The nuclei were counterstained with a DAPI staining solution (Service, Wuhan, China) for 10 min at room temperature, and TUNEL signals were observed with fluorescence microscopy (Olympus, Japan). Finally, at least ten visual fields were randomly selected per slice, and their apoptosis index (AI) was presented as the ratio of TUNEL-positive cells to total cells quantified using ImageJ software (National Institutes of Health [NIH], Bethesda, MD, USA).
H9C2 cell lines were obtained
from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and
routinely cultured in complete DMEM media containing 5 mM glucose and L-glutamine
(Hyclone, USA) supplemented with 10% fetal bovine serum (Gibco Laboratories,
USA) and 1% antibiotics (Gibco Laboratories, USA). The cells were cultured at 37
For cell transfection, single-gene si-RNA kits (RIBOBIO, Guangzhou, China) were mixed with Lipofectamine 2000 (GLPBIO, USA) in Opti-MEM (Gibco, USA) and transfected into H9c2 cells according to the manufacturer’s instructions. Each experiment was performed at least three times.
Cell viability was determined using a CCK-8
Assay Kit (Jiancheng, Nanjing, China). Briefly, H9c2 cells were cultured and
treated in 96-well plates, after which 10
LDH in cultured media was measured using a colorimetric assay kit (Jiancheng, Nanjing, China) according to the manufacturer’s instructions. Absorbance was spotted with the Perkin Elmer Microplate reader (EnSight PerkinElmer Victor 1420, USA).
To observe live and dead cells in the culture
dish concurrently, the Calcein-AM/PI assay (DOJINDO, Japan) was employed
according to the manufacturer’s instructions. In brief, H9c2 cells were loaded
with calcein-AM and PI regents at 37
Cardiac or cell protein per group
was extracted and separated on a 10% Tris
Cardiac tissue samples from the in vivo experiment were fixed with 4% paraformaldehyde and paraffin and sliced into sections. For myocardial histomorphology observation, these sections were counterstained with Hematoxylin and Evans Blue (HE). Some sections per group were subjected to immunofluorescence (IF) with an anti-PER2 antibody (1:50, BD, 611138) to examine PER2’s distribution in the heart.
In the H9c2 cell experiment, sterile coverslips were plated in 6-well plates,
and 0.5~1
All data are expressed as the Mean
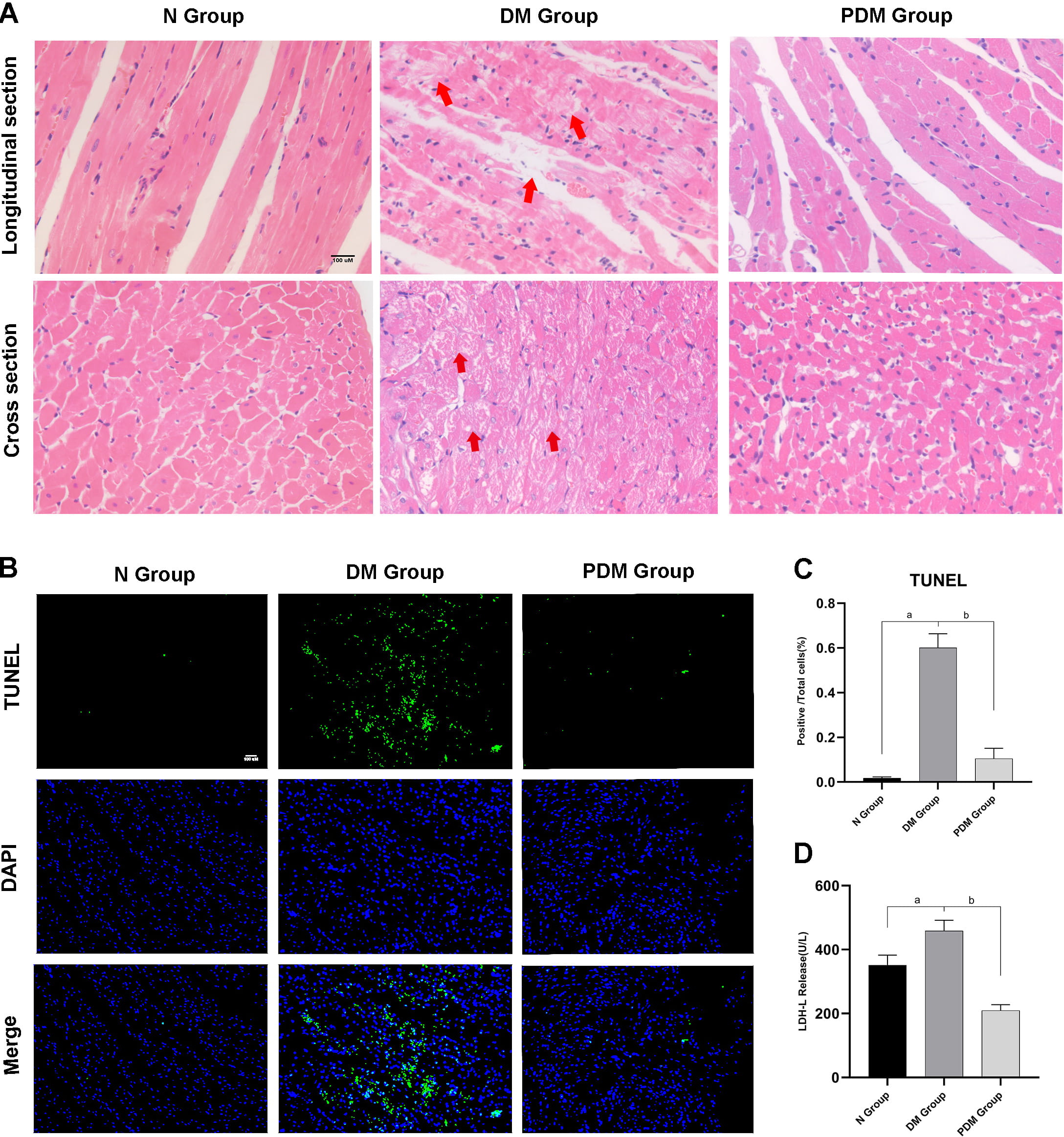
Compared to age-matched non-diabetic rats (N Group), diabetic rats (DM Group) exhibited obvious diabetic symptoms, such as high glucose, polydipsia, polyphagia, polyuria, weight loss, sour litter, and more. General characteristics, including body weight, heart weight, water intake, food intake, and FBG, were significantly altered in STZ-induced diabetic rats compared to their control counterparts (Fig. 1B). For cardiac function, diabetic rats displayed lower LVEF and LVFS and higher IVSs than non-diabetic rats (Fig. 1C). Furthermore, biochemical measurements registered substantially elevated MDA levels and a drop in SOD activity in the DM Group relative to the N Group (Fig. 1D). Myocardial morphology assessments (HE) established a series of myocardial damages, including disordered myocardial fibers and irregular and even dissolved vacuolated myocardiocytes with interstitial edema, in the DM Group compared to the N Group (Fig. 2A). Simultaneously, the apoptosis rate quantifying myocardial injury was higher in the DM Group than in the N Group (Fig. 2B,C).
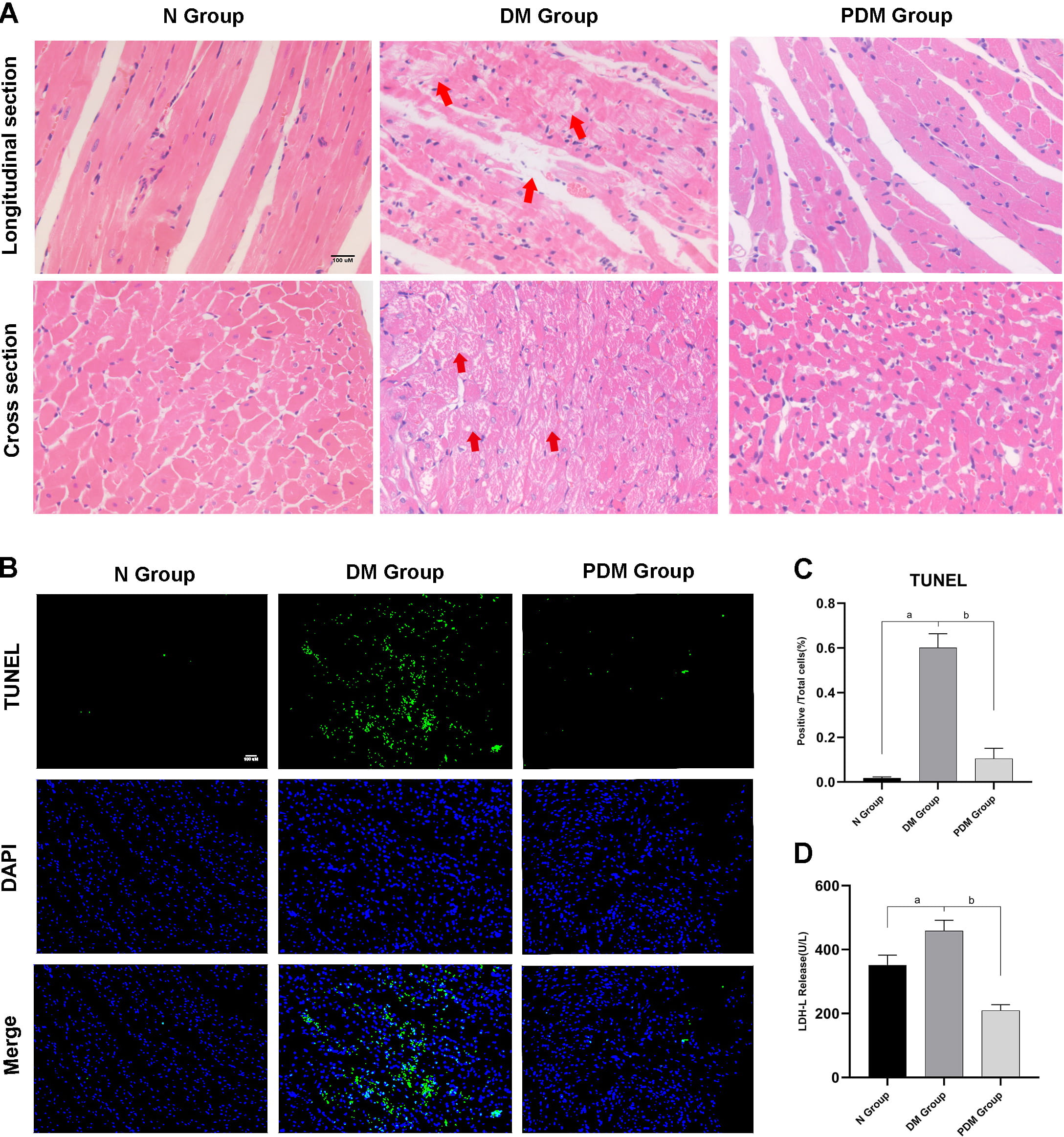 Fig. 2.
Fig. 2.Observation and quantification of cardiac injuries. (A)
HE pictures showing myocardial histomorphological changes of different groups
from its longitudinal and cross section (magnification
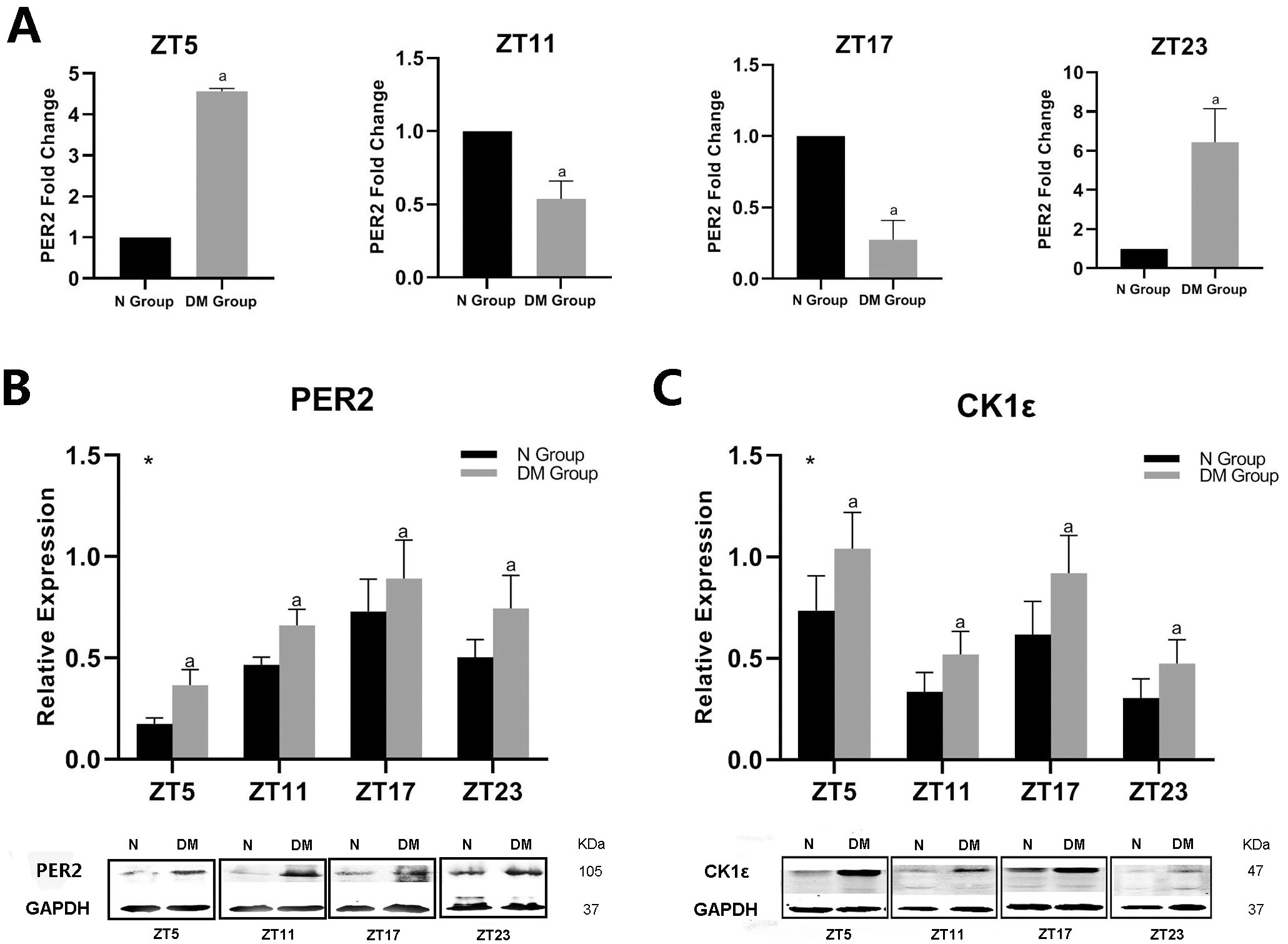
This study showed that diabetes markedly influenced the expression of PER2 and
CK1
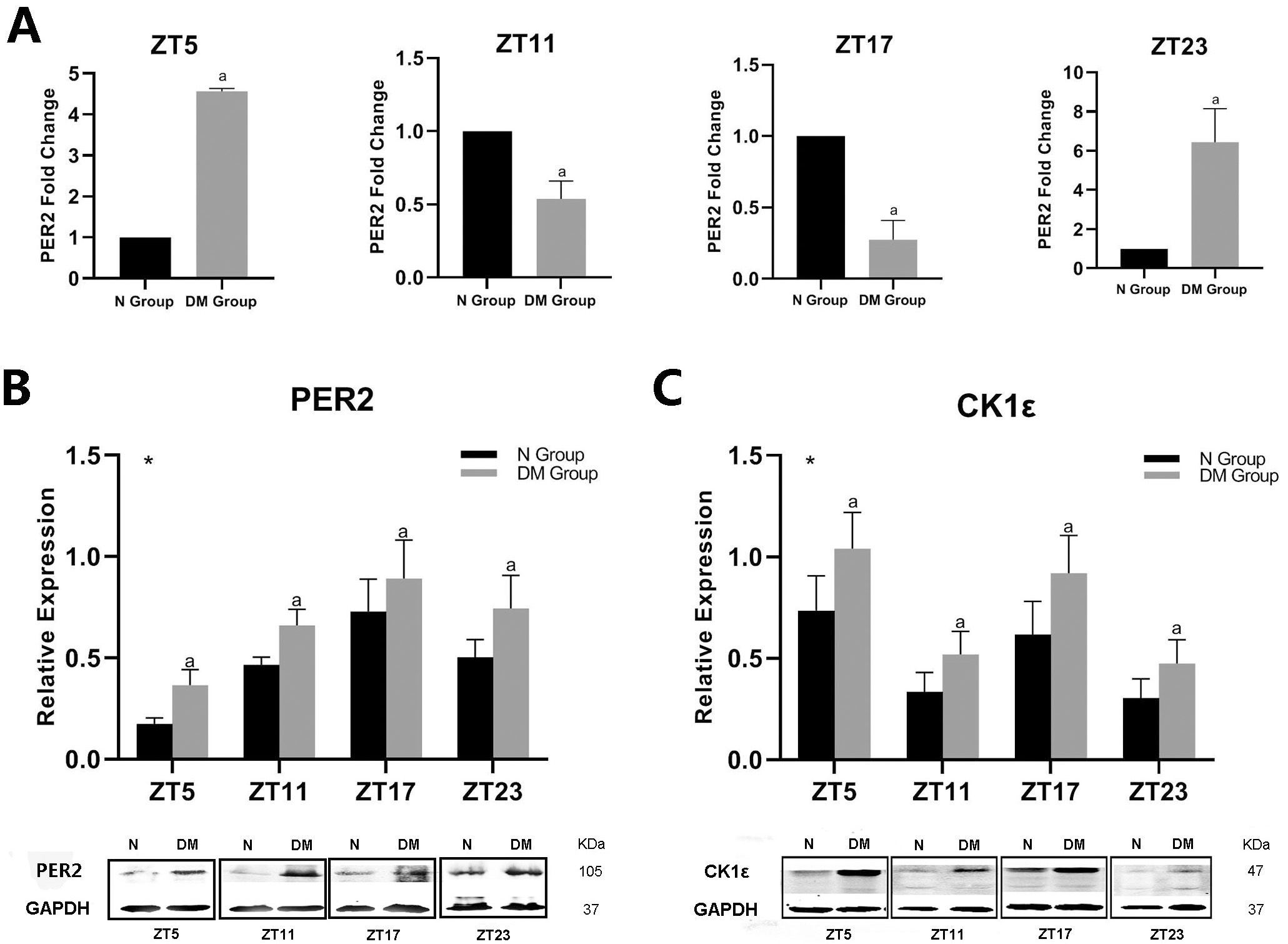 Fig. 3.
Fig. 3.Effect of diabetes on PER2 and CK1
| Protein | Groups | Amplitude | Phase (h) | p-value |
| PER2 | N Group | 17.0518 | 23.7 | 0.000458 |
| DM Group | 16.2469 | 24.1 | 0.005074 | |
| CK1 |
N Group | 17.1454 | 23.8 | 0.000029 |
| DM Group | 17.184 | 23.8 | 0.000024 | |
| When quantification of PER2 and CK1 | ||||
We measured the protein levels of CK1
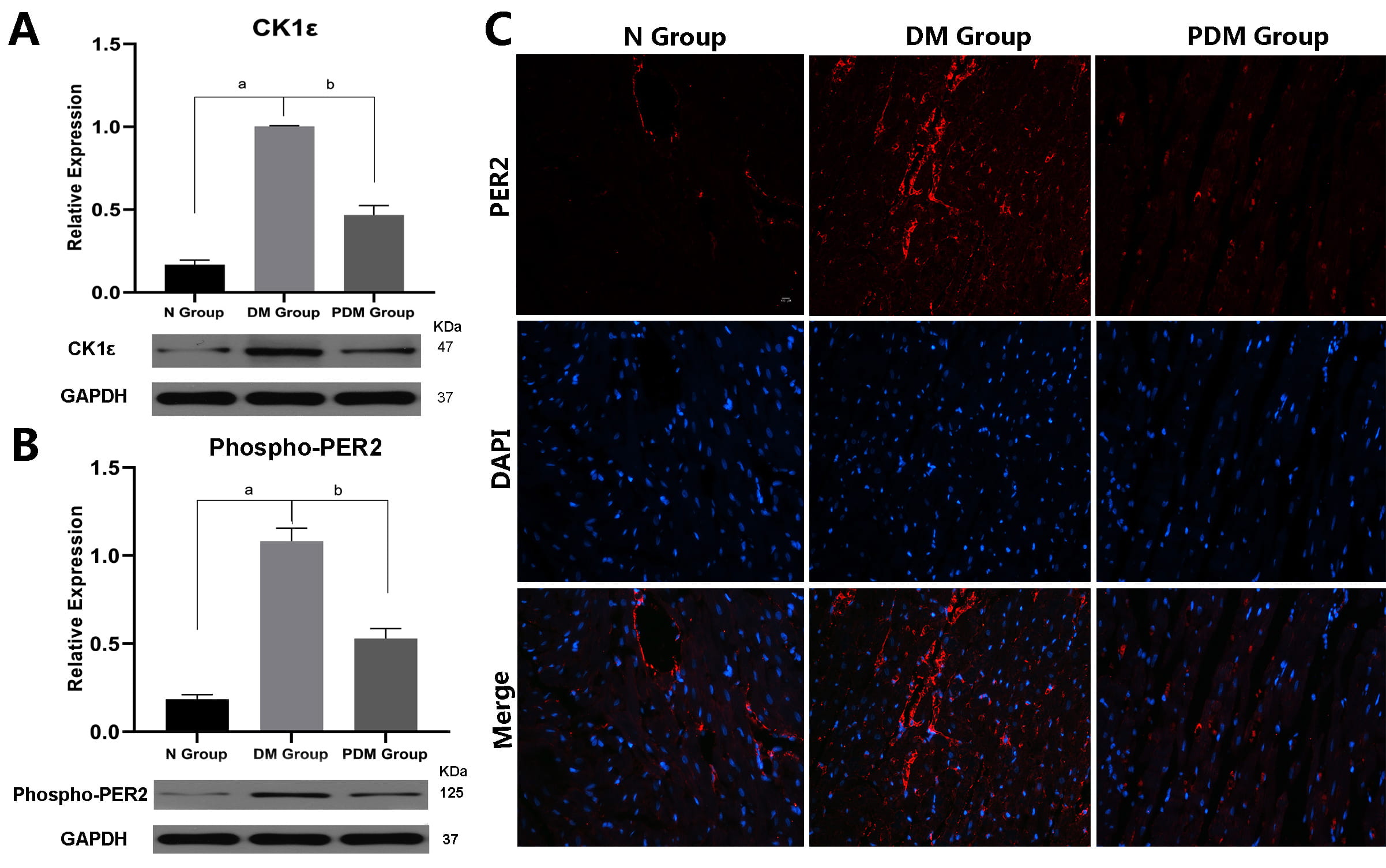 Fig. 4.
Fig. 4.PF-670462 remedied the expression of CK1
After treating diabetic rats with PF-670462 (PDM Group), we observed that their hair and cages became slightly dry and clean, and their activity levels improved spontaneously compared to diabetic rats (DM Group). While diabetes-caused myocardial structural damage remained notable in the PDM group, there were no longer dense masses of vacuolated cardiomyocytes and dissolved myocardial fibers (Fig. 2A). Also, the apoptosis rate and the biomarker of myocardial injury, LDH, dwindled in the PDM Group relative to the DM Group (Fig. 2B,C,D).
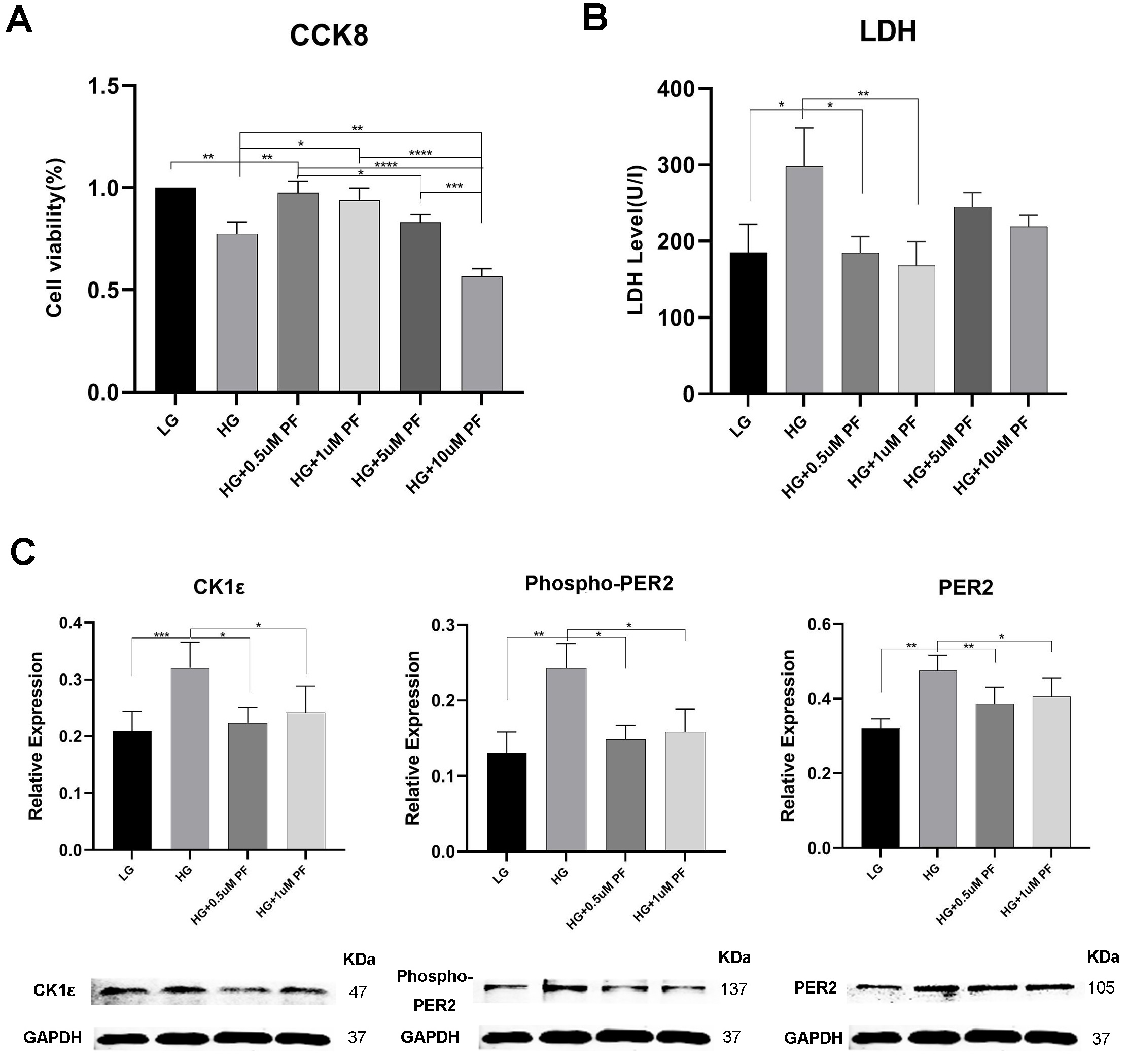
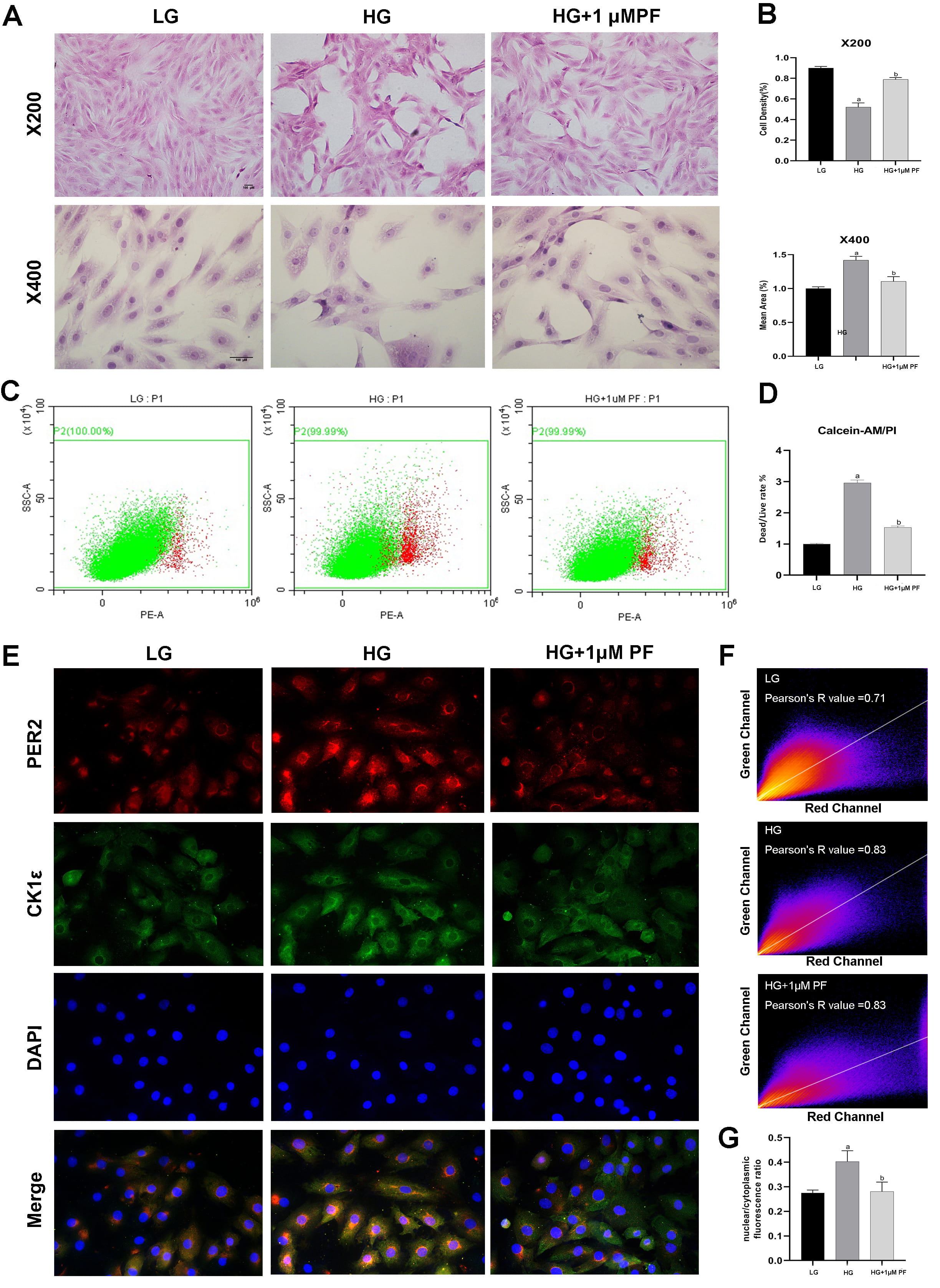
Consistent with our previous experiments [32, 33, 34], HG prompted a decrease in cell
viability and an increase in LDH release and the dead/live rate compared to the
LG group (Figs. 5A,B, 6C,D). Evaluations of H9c2 cells with HE highlighted a more
hypertrophic cell shape and lower cell density in the HG Group than in the LG
group (Fig. 6A and B). Additionally, CK1
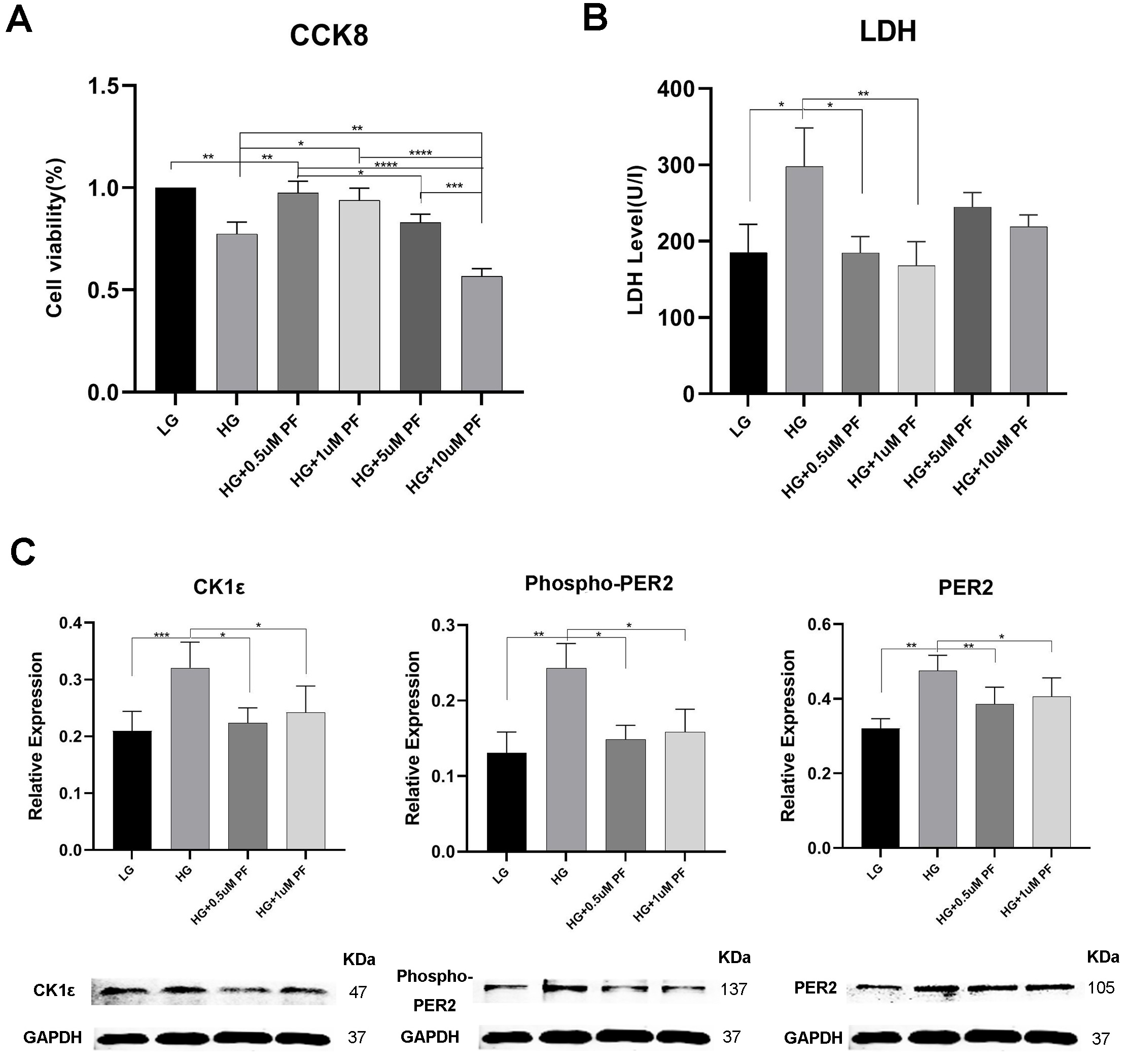 Fig. 5.
Fig. 5.The effect of PF-670462 on PER2 and CK1
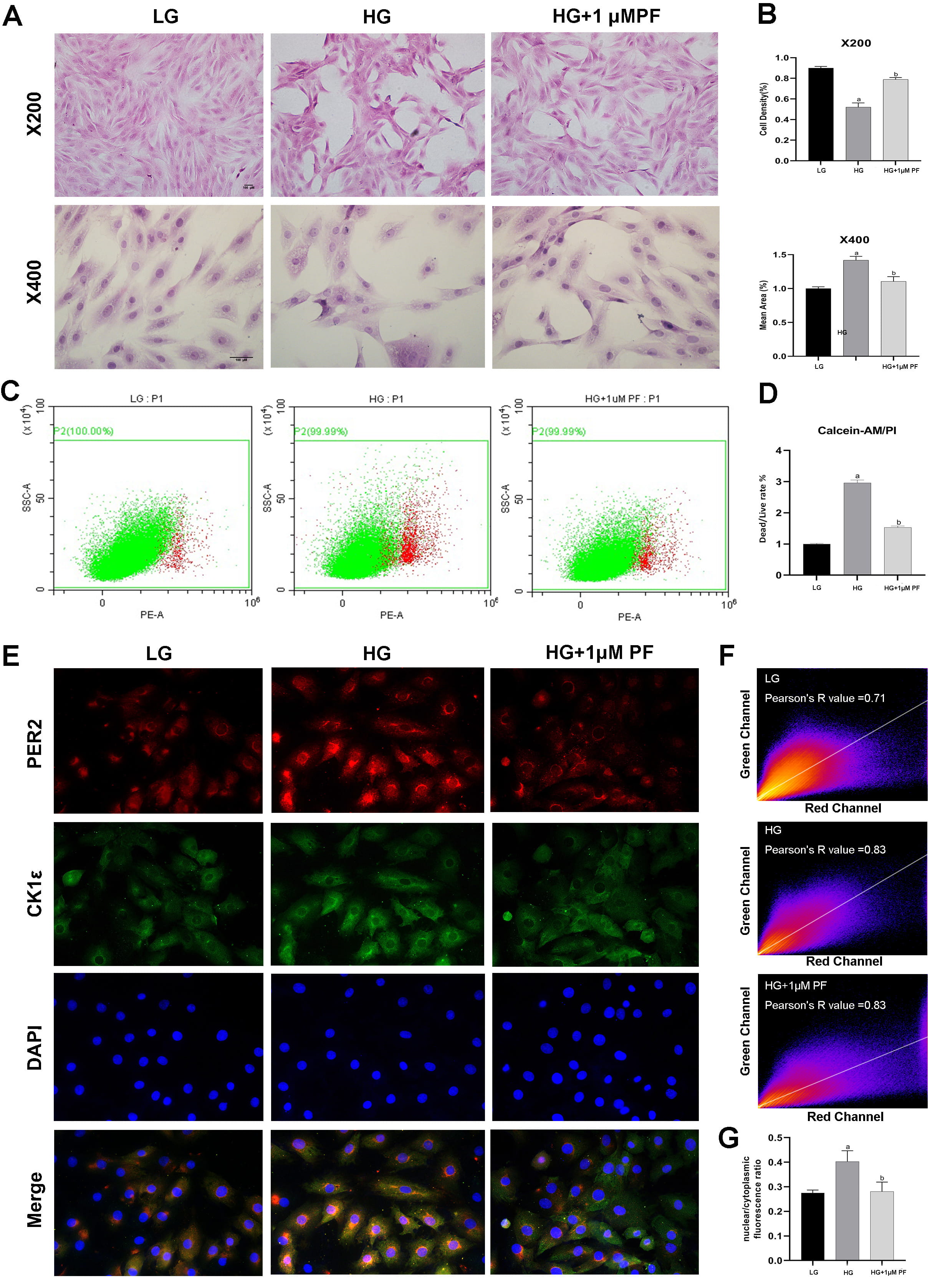 Fig. 6.
Fig. 6.Assessment of cell injuries and distributions of PER2 in H9c2.
(A) and (B) Repressenting respectively observation and quantification of
histomorphological structure of different groups in H9c2 (magnification
As shown in Fig. 6E, CK1
Exposing H9c2 cells to high glucose and treating them with 0.5
H9c2 cell treatment with PF-670462 in high glucose conditions saw a fall in the
expression of CK1
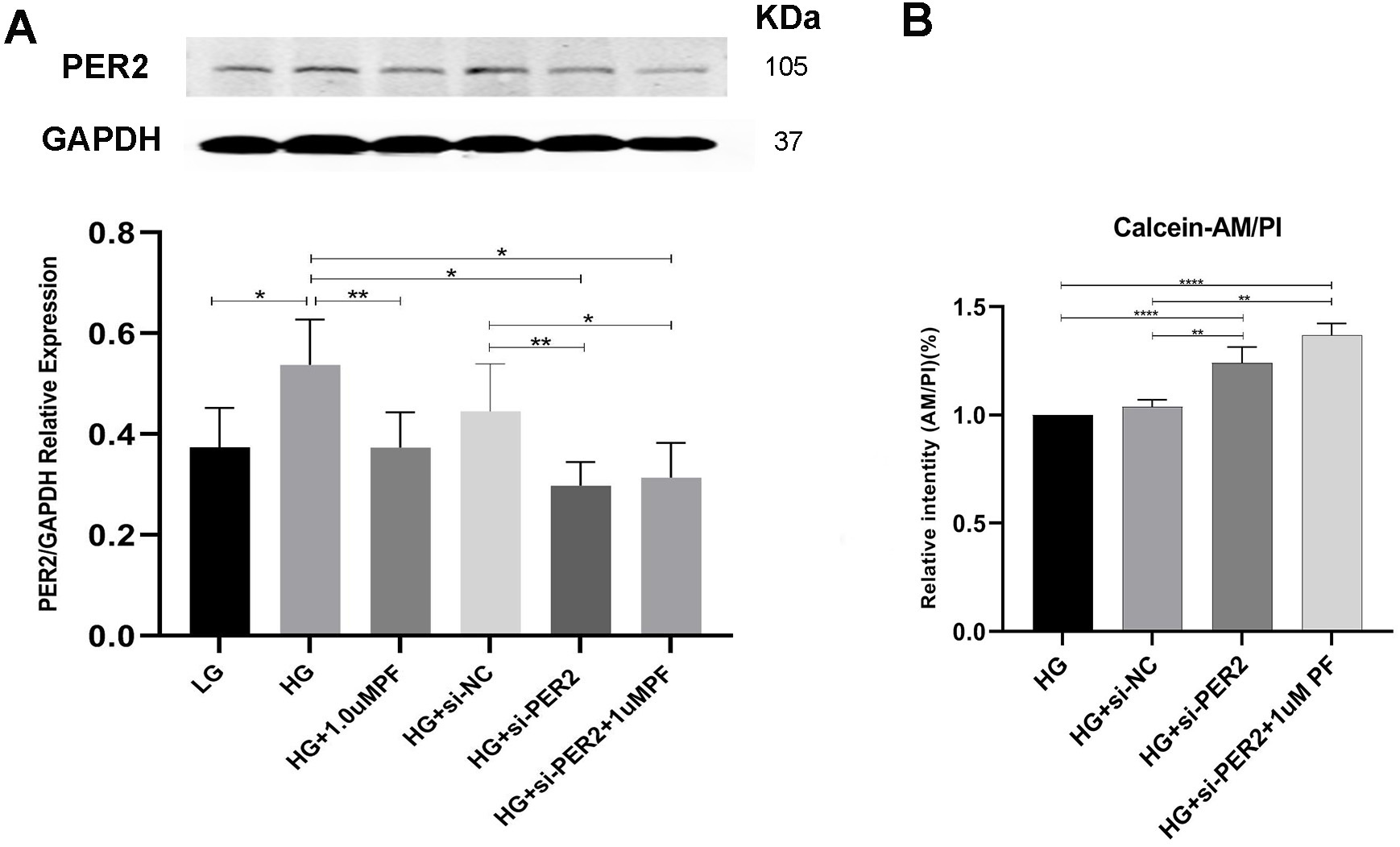
As shown in Fig. 7A and B, knocking down PER2 in the presence of high glucose resulted in improved cell viability relative to the controls. PF-670462’s attempt to downregulate PER2 and improve cell viability in high glucose conditions beyond the rates achieved by PER2 knockdown was comparable to that of HG+si-PER2 (Fig. 7A and B).
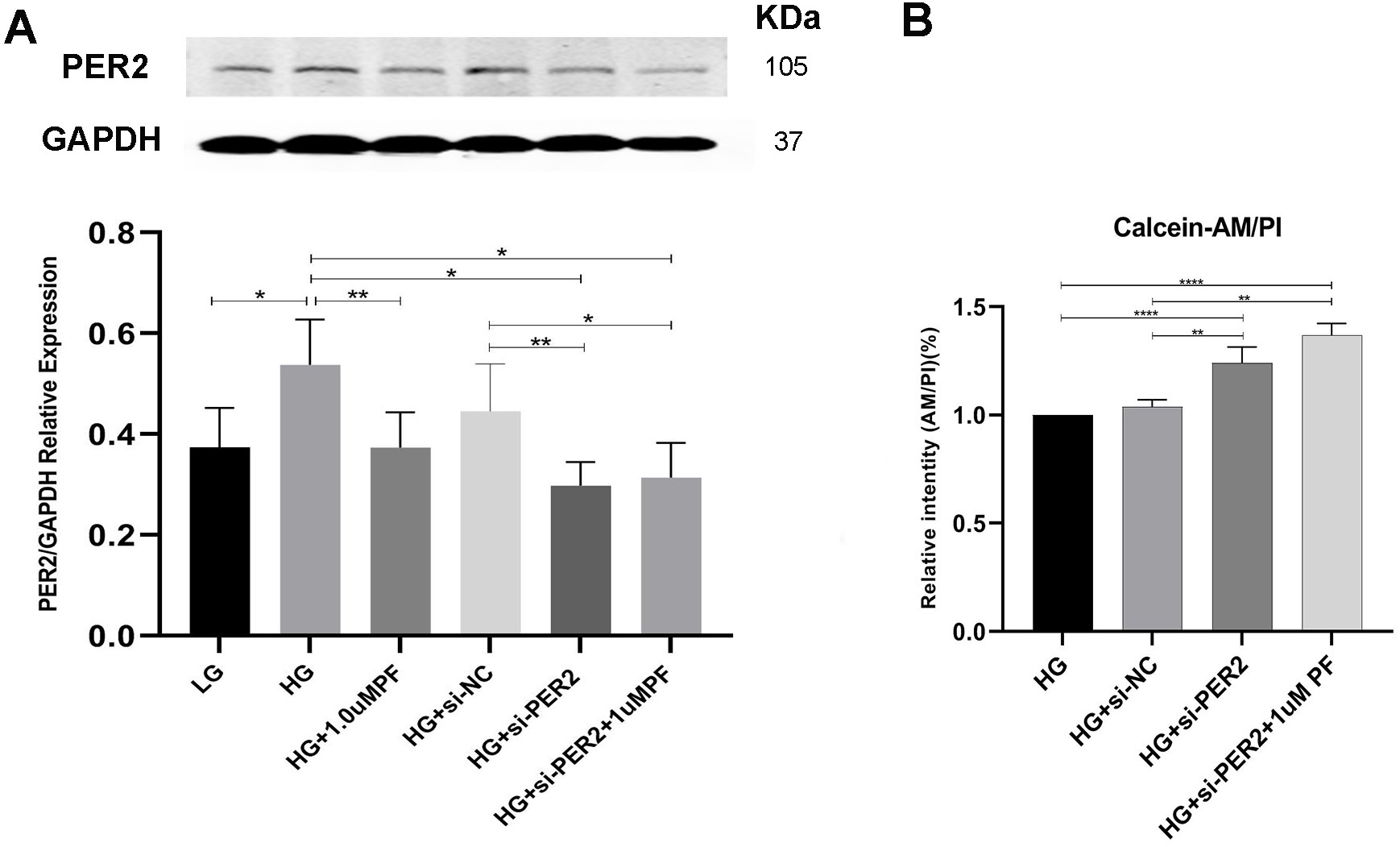 Fig. 7.
Fig. 7.Pharmacological and genetic targeting of PER2 in high-glucose
condition. (A) Quantification of protein expression of PER2 treated with
PF-670462 or siRNA in high-glucose condition. (B) Relative intentity of
Calcein-AM/PI detected by fluorescent Microplate reader. Data was expressed as
the Mean
This investigation contributes to growing evidence that molecular clockwork is
compromised during diabetes. It revealed that diabetes resets PER2’s oscillation
in association with activated CK1
Diabetes mellitus is now one of the most prevalent disorders, and it could lead
to devastating myocardial injuries on the cardiac structure and function [1, 2, 3].
Cardiovascular disease (CVD) is the leading cause of death in type-1 and type-2
diabetes, and, while
Although some studies have reported the impact of diabetes on PER2 [40, 41, 42, 43], a clear understanding of the extent of its influence and its potential mechanism must still be explored. Consistent with what other studies have demonstrated [42, 43, 44], we found that diabetes altered PER2 expression at the mRNA level at different time points (Fig. 3A). Investigations have shown that the PER2 protein acts as a remarkable functional regulator through its cyclic accumulation and degradation in mammals [14, 15, 16]. Based on this, we examined the oscillation of the PER protein and established that diabetes changed PER’s stability, with its phase delayed and amplitude decreased (Fig. 3B). The stability of PER2 depends on its phosphorylation status, and the rivalry between two unique competing sites on the PER2 protein is the key step in the PER2 circadian phosphoswitch that determines its period.
There is proof that CK1
Targeting CK1
As demonstrated in previous studies, PF-670462 slows down PER protein turnover and lengthens the circadian period in normal animals [21, 29, 30, 31]. Currently, the number of investigations reporting that the impact of PF-670462 relies on the status of PER2 being influenced by metabolic and environmental stimuli are mounting [12, 24, 46, 47, 48]. In our research, PF-670462 exhibited no influence on PER2 expression and cell injury when PER2 was knocked down in the presence of high glucose (Fig. 7A and B), suggesting that PF-670462 aids high-glucose-engendered injuries only when the PER2 protein is upregulated, consistent with a report that PF-670462 inhibits IgE-mediated allergic reactions only when PER2 is increased [48].
Despite its promising revelations, our study had several limitations. Due to
financial constraints, we have not used PER2
In summary, this work first provides additional evidence that diabetes
compromises PER2 in association with activated CK1
ZYX, SL and QH designed the research study. QH and MJ performed the research. QH and ZQ analyzed the data and wrote the manuscript. ZYX, SL, BZ, MJ, ZQ, and QH contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
All institutional and national guidelines for the care and use of laboratory animals were followed. The protocols used on all the animals have been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Renmin Hospital of Wuhan University (#20180214).
The authors would like to thank the Central Laboratory of Remin Hospital of Wuhan University for the expert technical assistance.
This study was supported by grants from National Natural Science Foundation of China [grant number: 81671891] and [grant number: 81901947]. The organization had no role in the design of the study, the collection, analysis, and interpretation of data and writing the manuscript.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://www.imrpress.com/journal/FBL/27/2/10.31083/j.fbl2702058.