In assisted reproductive technology (ART), hormone replacement treatment embryo transfer (HRT-ET) is common. However, the appropriate serum progesterone concentration is not well known in the case of transvaginal progesterone administration. The purpose of the current study is to investigate the relationship between serum progesterone concentration and pregnancy rate in HRT-ET using progesterone vaginal tablets. This is a retrospective cohort study, the authors investigated all patients who underwent HRT-ET at this hospital for the first time from September 1, 2010 to September 30, 2015. A total of 227 cycles were performed in all 123 patients who underwent HRT-ET. In the total 123 cycles of first time HRT-ET, 32 cycles (26.0%) developed a pregnancy. The results of the multivariate analysis indicated that the patient’s age, embryo grade, serum estradiol concentration, and progesterone concentration had significant differences in relation to the pregnancy rate (p < 0.05). The latter rate tends to be lower when the serum progesterone concentration is less than 5 or more than 15 ng/ml. Therefore, the authors suggest that the appropriate serum progesterone concentration ranges from 5 to 15 ng/ml in HRT-ET using progesterone vaginal tablets.
According to the data from the International Committee for Monitoring Assisted Reproductive Technologies (ICMART), approximately 1.5 million assisted reproductive technology (ART) cycles are performed and 350,000 babies are born annually [1].
In ART, embryo freezing is performed for the purpose of avoiding embryo transfer (ET) under a non-physiological endocrine environment after ovarian stimulation, preventing ovarian hyperstimulation syndrome (OHSS) or preserving surplus embryos [2]. As a method for thawing the embryo transfer, there are two major protocols: natural cycle ET (natural-ET) and hormone replacement treatment ET (HRT-ET). In natural-ET, the present authors check the ovulation timing in the patient’s menstrual cycle and perform ET according to the embryo’s age, (e.g., a Day 3 embryo is transferred 3 days after ovulation). In this hospital, HRT-ET is selected, for example, when the patient has an imbalance of hormones such as irregular menstruation and repeatedly unsuccessful attempts with natural-ET.
In HRT-ET, supplemental estradiol and progesterone are required because ovulation and corpus luteum formation do not occur and endogenous hormones are likely inadequate. Estradiol has the effect of thickening the uterine endometrium, and progesterone has the effect of endometrial gland formation and endometrium condition adjustment for trophoblast invasion [3, 4].
Progesterone can be administered by oral, intramuscular, or vaginal routes [5]. However, the appropriate serum progesterone concentration of each administration is controversial. Following spontaneous ovulation, the midluteal serum progesterone concentration is typically more than 10 ng/ml [6]. Therefore, in general, the appropriate serum progesterone concentration for pregnancy is > 10 ng/ml.
Worldwide, transvaginal administration is the standard route [7, 8]. Although serum progesterone concentration in transvaginal administration is lower than other modes of administration, it is known to exhibit good pregnancy rates [9-11]. It is also known that progesterone administered directly to the vagina acts on the uterus before being metabolized in the liver, resulting in an eight-fold increase in endometrial progesterone concentration compared with intramuscular administration, despite a three-fold decrease in serum progesterone concentration [12]. One of the latest studies from Australia suggested that the implantation rate is reduced by a higher serum progesterone concentration and that the appropriate serum progesterone concentration is from 70 to 90 nmol/l (approximately from 22.0 to 28.3 ng/ml) in the case of vaginal pessary administration [13]. Minimal research has been conducted regarding the appropriate serum progesterone concentration for pregnancy in the case of transvaginal progesterone administration.
The purpose of the current study was to investigate the relationship between serum progesterone concentration and the pregnancy rate with transvaginal administration.
This was a retrospective cohort study at St. Luke’s International Hospital, Tokyo, Japan. The authors investigated all patients who underwent a thawed ET in hormone replacement cycle at this hospital for the first time from September 1, 2010 to September 30, 2015. The authors excluded patients who underwent frozen ET transfer for the second or greater time.
In this hospital, the HRT-ET protocol is as follows (Figure 1): from day 1 to 3 of the menstrual cycle, the authors evaluate the patient’s condition to evaluate abnormalities of their ovaries, such as an unruptured corpus luteum cyst. Next, patients begin taking two sheets of 0.72 mg percutaneous absorption natural type estradiol product every other day from days 2 to 8 of the menstrual cycle. From days 10 to 12 of the menstrual cycle, the authors check their endometrial thickness and ovaries by transvaginal ultrasonography. When the endometrial thickness reaches 8 mm or greater and no large follicle is seen in their ovaries, they decide to carry out the ET. On day 14 of the menstrual cycle, the patients take six sheets. From day 16, they take three sheets every other day and begin administration two or three times a day of a progesterone vaginal tablet. They received ET according to the following schedule: day 2 embryo on day 17, day 3 embryo on day 18, and day 5 embryo on day 20. HRT was continued until approximately five weeks after menstruation to confirm the presence or absence of a pregnancy; in the case of pregnancy, HRT is continued until approximately eight weeks after menstruation. The primary outcome was pregnancy rate. The authors determined that pregnancy was established by acknowledging the gestational sac in the uterus by ultrasound examination. Other variables, including patients’ characteristics, endometrial condition, embryo condition, and hormonal levels, were obtained from the date of the HRT-ET. Among them, serum progesterone concentration was classified into four groups: <5, 5-9.9, 10.0-14.9, and 15 ≤ ng/ml. The thickness of the endometrium was measured by transabdominal ultrasound on the day of ET. The embryo grade was classified according to Veeck’s criteria [14], and the authors classified the embryo into three groups: group 1 as the blastocyst, group 2 as grades 1 and 2 of a day 2 and 3 embryo, and group 3 as grade 3 and 4 of a day 2 and 3 embryo (Figure 2). Assisted hatching (AH) is a reproductive technique using acidic Tyrode’s solution or applying laser photoablation to the zona pellucida of the embryo to help the embryo to hatch [15]. Some studies have reported that AH yields a higher implantation rate. The present authors used acidic Tyrode’s solution for AH. All data were extracted from electronic charts.
 Figure 1.
Figure 1.— ET indicates embryo transfer. Progesterone vaginal tablet† indicates progesterone tablet. Estrana tape® §indicates percutaneous absorption natural type estradiol product.
This diagram shows a typical HRT-ET protocol in our hospital. Estrana tape® § 0.72 mg is taken every other day from day 2 of menstrual cycle and the number of the sheets are adjusted as shown. Progesterone vaginal tablet† begins at 200-300 mg/day from day 16. Day 2 embryo is transferred on day 17. Day 3 embryo is transferred on day1 8. Day 5 embryo is transferred on day 20. Hormone replacement treatment is continued until about 5 weeks of menstruation that we can confirm the presence or absence of pregnancy.
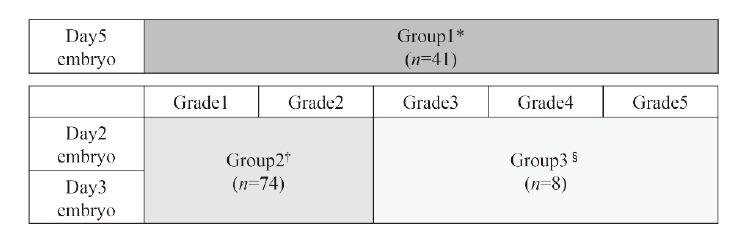 Figure 2.
Figure 2.— Day 5 embryos are classified into Group 1*. Grade 1 and 2 of Day 2 and 3 embryos are classified into Group 2†. Grades 3, 4, 5 of Day 2 and 3 embryos are classified into Group3§.
For univariate analyses, a chi-square test was applied to categorical data and a student’s t-test was applied to continuous data. Logistic regression was performed for multivariate analysis. All analyses were performed by SPSS Statistics version 19 and Stata IC version 11. St. Luke’s International Hospital Institutional Review Board approved this study.
A total of 227 cycles were performed in 123 patients who underwent HRT-ET. In the total 123 cycles of first time HRT-ET, 32 cycles (26.0%) developed a pregnancy. The mean age (± standard deviation [SD]) was 38.6 (± 4.1) years-old. The mean serum progesterone concentration (±SD) was 9.9 ng/ml (±4.3). The number of each embryo group was as follows: group 1, 41 subjects (16 cases developed a pregnancy), group 2, 75 subjects (16 cases developed a pregnancy), and group 3, seven subjects (0 cases developed a pregnancy). In five cycles of the 123 cycles, the authors transferred two embryos at the same time. In eight cycles of the 123 cycles, the authors used assisted hatching. The age of the patients, the serum estradiol concentration, and the serum progesterone concentration were obtained on the date of the HRT-ET. The progesterone serum concentration and the number and pregnancy rate are shown in Table 1 for each group.
| Serum progesterone concentration (ng/ml) | |||||
|---|---|---|---|---|---|
| < 5.0 (n=13) | 5.0 - 9.9 (n=55) | 10.0 - 14.9 (n=40) | 15 ≤ (n=15) | Total (n=123) | |
| Pregnancy, n (%) | 1 (7.7) | 18 (32.7) | 12 (30.0) | 1 (6.7) | 32 (26.0) |
We took the univariate analysis for the items compared with pregnancy or failure. The results of univariate analysis are as shown in Table 2. Patient age and embryo grade showed a significant difference in relation to the pregnancy rate (p < 0.05). The results of multivariate analysis are shown in Table 3. Patient age, serum estradiol concentration, embryo grade, and serum progesterone concentration showed significant differences in relation to the pregnancy rate (p < 0.05).
| Pregnancy (n = 32) | Non-pregnancy (n = 91) | Total (n = 123) | p-value | |
|---|---|---|---|---|
| Age, years (SD) | 36.6 (3.4) | 39.3 (4.1) | 38.6 (4.1) | <0.01 |
| Endometrial thickness, mm (SD) | 9.4 (2.4) | 9.5 (2.6) | 9.4 (2.5) | 0.94 |
| Progesterone, ng/ml (SD) | 9.9 (3.6) | 9.9 (4.6) | 9.9 (4.3) | 0.99 |
| Estradiol, pg/ml (SD) | 309.6 (240.4) | 244.9 (166.4) | 261.7 (189.5) | 0.17 |
| Grade of embryo, n (%) | 0.04 | |||
| Group 1 | 16 (50.0) | 25 (27.5) | 41 (33.3) | |
| Group 2 | 16 (50.0) | 59 (64.8) | 75 (61.0) | |
| Group 3 | 0 (0.0) | 7 (7.7) | 7 (5.7) | |
| Two embryo transfers, n (%) | 0 (0.0) | 5 (5.5) | 5 (4.1) | 0.33 |
| Assisted hatching, n (%) | 2 (6.3) | 6 (6.6) | 8 (6.5) | 1.00 |
| Odds ratio | 95% confidence interval | p-value | |
|---|---|---|---|
| Age, year | 0.84 | 0.74 - 0.95 | < 0.01 |
| Estradiol, 100pg/ml | 1.41 | 1.06 - 1.87 | 0.02 |
| Grade of embryo | 2.39 | 1.00 - 5.72 | < 0.05 |
| Progesterone, ng/ml | < 0.05 | ||
| < 5.0 | 0.09 | 0.01 - 1.34 | 0.08 |
| 5.0 - 9.9 | 0.75 | 0.27 - 2.05 | 0.58 |
| 10.0 - 14.9 | Reference | ||
| ≥ 15.0 | 0.04 | 0.01 - 0.48 | 0.01 |
The present results suggested that the appropriate serum progesterone concentration in the case of transvaginal administration would be from 5 to 15 ng/ml. The results also suggested that serum estradiol concentrations showed a significant difference with pregnancy rate. However, there are negative reports regarding the relationship between serum estradiol concentrations on the date of ET and the pregnancy rate [16]; at present, the relationship is controversial and further studies are necessary.
There is a report that included cases with a serum progesterone concentration > 20 ng/ml on ET, showing higher pregnancy rates than cases with less than 20 ng/ml in the donor oocytes cycle [17]. Conversely, in another report, a serum progesterone concentration of > 20 ng/ml on ET showed a lower rate of live births and a higher rate of pregnancy losses [18]. However, these reports used intramuscular progesterone administration.
There are two recent studies that examined cases with transvaginal administration. One is mentioned above, and the authors suggested that an appropriate serum progesterone concentration is from 70 to 90 nmol/l in HRT-ET and that a higher progesterone concentration [≥100 nmol/l (31.4 ng/ml)] is associated with a reduced implantation rate [13]. Another study compared doses of progesterone (300 mg and 600 mg) for luteal phase support with intrauterine insemination (IUI). Because there was no significant difference in the pregnancy rate, they concluded that 300 mg of intravaginal progesterone is the lowest effective dose and is the maximum dose of luteal phase support in IUI [19]. However, the study showed no significant difference in serum progesterone concentration between 300 mg and 600 mg on hCG injection day. Therefore, the appropriate serum progesterone concentration in the case of transvaginal administration in IUI is still unknown.
In human menstrual cycles, the uterine endometrium undergoes changes from the proliferative to the secretory phase after ovulation. Uterine sensitivity to implantation in the secretory phase is classified as pre-receptive, receptive, and refractory phases. During the secretory phase, the uterus is considered pre-receptive from one to seven days following ovulation; it then becomes receptive during the mid-secretory phase, which spans from seven to ten days after ovulation. The refractory phase comprises the remainder of the secretory phase [20]. There is a study that reported that the abortion rate increases if the time of implantation is later than ten days after ovulation [21].
In the case of a serum progesterone concentration of less than 5 ng/ml, the present authors consider the possibilities that vaginal administration was not performed accurately by each patient and that the absorption of the medication was low. A possible explanation is that the endometrium was not sufficiently developed to support implantation.
In the case of a serum progesterone concentration of more than 15 ng/ml, the present data showed a lower pregnancy rate. One idea to explain this situation is that a higher serum progesterone concentration may lead to a direct effect on the “pre-receptive and receptive window” in the endometrium by shifting it earlier in time. Accelerated endometrial development could de-synchronize uterine and embryo development and lead to decreased implantation or a “poor quality” implantation [8]. In addition, vaginal progesterone administration can provide a higher endometrial progesterone concentration than other modes of administration [9]. Therefore, the present authors consider that the patient whose serum progesterone concentration is more than 15 ng/ml on the date of ET may have excessive uterine endometrial development and their pregnancy rate may be lower with vaginal progesterone administration.
A hypothesis that explains why the progesterone serum concentration becomes higher is that there might be ovulation of the patient’s own hidden follicular development and endogenous progesterone is secreted. If hidden early ovulation occurs, the endometrium is exposed to a higher concentration of progesterone over a longer period. In this case, the de-synchronization of the uterine membrane and the embryo state could occur.
In the current study, there was a statistically significant difference between the pregnancy rate and serum progesterone concentration in HRT-ET. The present study suggested that the appropriate serum progesterone concentration in the case of vaginal administration in HRT-ET would range from 5 to 15 ng/ml. However, this is a small study at one facility. Further studies will be necessary to investigate the universality of the appropriate serum progesterone concentration in HRT-ET.
